Ranexa
Generic name: ranolazine
Drug class: Antianginal agents
Medically reviewed by A Ras MD.
What is Ranexa?
Ranexa is a prescription medicine used to treat angina that keeps coming back (chronic angina). Ranexa may be used with other medicines that are used for heart problems and blood pressure control.
It is not known if Ranexa is safe and effective in children.
Description
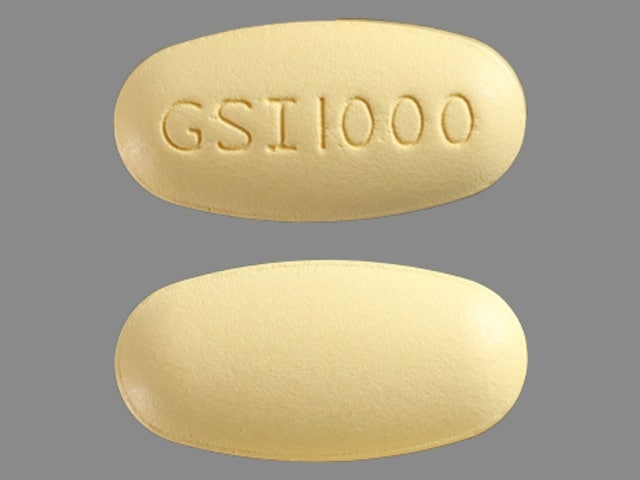
Ranexa (ranolazine) is available as a film-coated, non-scored, extended-release tablet for oral administration.
Ranolazine is a racemic mixture, chemically described as 1-piperazineacetamide, N-(2,6-dimethylphenyl)-4-[2-hydroxy-3-(2-methoxyphenoxy)propyl]-, (±)-. It has an empirical formula of C24H33N3O4, a molecular weight of 427.54 g/mole, and the following structural formula:

Ranolazine is a white to off-white solid. Ranolazine is soluble in dichloromethane and methanol; sparingly soluble in tetrahydrofuran, ethanol, acetonitrile, and acetone; slightly soluble in ethyl acetate, isopropanol, toluene, and ethyl ether; and very slightly soluble in water.
Ranexa tablets contain 500 mg or 1000 mg of ranolazine and the following inactive ingredients: carnauba wax, hypromellose, magnesium stearate, methacrylic acid copolymer (Type C), microcrystalline cellulose, polyethylene glycol, sodium hydroxide, and titanium dioxide. Additional inactive ingredients for the 500 mg tablet include polysorbate 80 and FD&C Yellow No. 6 Lake; additional inactive ingredients for the 1000 mg tablet include lactose monohydrate, triacetin, and Iron Oxide Yellow.
Mechanism of Action
The mechanism of action of ranolazine’s antianginal effects has not been determined. Ranolazine has anti-ischemic and antianginal effects that do not depend upon reductions in heart rate or blood pressure. It does not affect the rate-pressure product, a measure of myocardial work, at maximal exercise. Ranolazine at therapeutic levels can inhibit the cardiac late sodium current (INa). However, the relationship of this inhibition to angina symptoms is uncertain.
The QT prolongation effect of ranolazine on the surface electrocardiogram is the result of inhibition of IKr, which prolongs the ventricular action potential.
Who should not take Ranexa?
Do not take Ranexa if:
- you take any of the following medicines:
- for fungus infection: ketoconazole (Nizoral), itraconazole (Sporanox, Onmel)
- for infection: clarithromycin (Biaxin)
- for depression: nefazodone
- for HIV: nelfinavir (Viracept), ritonavir (Norvir), lopinavir and ritonavir (Kaletra), indinavir (Crixivan), saquinavir (Invirase)
- for tuberculosis (TB): rifampin (Rifadin), rifabutin (Mycobutin), rifapentine (Priftin)
- for seizures: phenobarbital, phenytoin (Phenytek, Dilantin, Dilantin-125), carbamazepine (Tegretol)
- St. John’s wort (Hypericum perforatum)
- you have scarring (cirrhosis) of your liver
What should I tell my healthcare provider before taking Ranexa?
Before you take Ranexa, tell your doctor if you:
- have or have a family history of a heart problem, called ‘QT prolongation’ or ‘long QT syndrome’.
- have liver problems.
- have kidney problems.
- are pregnant or plan to become pregnant. It is not known if Ranexa will harm your unborn baby.
- are breast-feeding or plan to breast-feed. It is not known if Ranexa passes into your breast milk. You and your doctor should decide if you will breast-feed.
Tell your doctor about all the medicines you take, including all prescription and nonprescription medicines, vitamins, and herbal supplements. Ranexa may affect the way other medicines work and other medicines may affect how Ranexa works.
Tell your doctor if you take medicines:
- for your heart
- for cholesterol
- for diabetes
- for infection
- for fungus
- for transplant
- for nausea and vomiting because of cancer treatments
- for mental problems
Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine.
How should I take Ranexa?
- Take Ranexa exactly as your doctor tells you.
- Your doctor will tell you how much Ranexa to take and when to take it.
- Do not change your dose unless your doctor tells you to.
- Tell your doctor if you still have symptoms of angina after starting Ranexa.
- Take Ranexa by mouth, with or without food.
- Swallow the Ranexa tablets whole. Do not crush, break, or chew Ranexa tablets before swallowing.
- If you miss a dose of Ranexa, wait to take the next dose of Ranexa at your regular time. Do not make up for the missed dose. Do not take more than 1 dose at a time.
- If you take too much Ranexa, call your doctor, or go to the nearest emergency room right away.
What should I avoid while taking Ranexa?
- Grapefruit and grapefruit juice. Limit products that have grapefruit in them. They can cause your blood levels of Ranexa to increase.
- Ranexa can cause dizziness, lightheadedness, or fainting. If you have these symptoms, do not drive a car, use machinery, or do anything that needs you to be alert.
What are the possible side effects of Ranexa?
Ranexa may cause serious side effects, including:
- changes in the electrical activity of your heart called QT prolongation. Your doctor may check the electrical activity of your heart with an ECG. Tell your doctor right away if you feel faint, lightheaded, or feel your heart beating irregularly or fast while taking Ranexa. These may be symptoms related to QT prolongation.
- kidney failure in people who already have severe kidney problems. Your doctor may need to do tests to check how your kidneys are working.
The most common side effects of Ranexa include:
- dizziness
- headache
- constipation
- nausea
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of Ranexa. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Ranexa
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not use Ranexa for a condition for which it was not prescribed. Do not give Ranexa to other people, even if they have the same condition you have. It may harm them.
The Patient Information summarizes the most important information about Ranexa. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Ranexa that is written for health professionals.
For more information call Gilead Sciences, Inc. at 1-800-445-3235.
How should I store Ranexa?
Store Ranexa tablets at room temperature between 59° to 86°F (15° to 30°C).
Keep Ranexa and all medicines out of the reach of children.
What are the ingredients in Ranexa?
Active ingredient: ranolazine
Inactive ingredients:
500 mg tablet: carnauba wax, hypromellose, magnesium stearate, methacrylic acid copolymer (Type C), microcrystalline cellulose, polyethylene glycol, sodium hydroxide, titanium dioxide, polyvinyl alcohol, talc, Iron Oxide Yellow, and Iron Oxide Red.
1000 mg tablet: carnauba wax, hypromellose, magnesium stearate, methacrylic acid copolymer (Type C), microcrystalline cellulose, polyethylene glycol, sodium hydroxide, titanium dioxide, lactose monohydrate, triacetin, and Iron Oxide Yellow.
Label
PRINCIPAL DISPLAY PANEL – 500 mg Label
- NDC 61958-1001-1
60 tablets - Ranexa®
(ranolazine extended-
release tablets) - 500 mg
- Rx only


PRINCIPAL DISPLAY PANEL – 1000 mg Label
- NDC 61958-1002-1
60 tablets - Ranexa®
(ranolazine extended-
release tablets) - 1000 mg
- Rx only

SRC: NLM .
