Xeljanz
Generic name: tofacitinib
Brand names: Xeljanz, Xeljanz XR
Dosage form: oral extended-release tablet, oral tablet, oral solution
Drug class: Antirheumatics
Medically reviewed by A Ras MD.
What is Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
Xeljanz, Xeljanz XR and Xeljanz Oral Solution is a prescription medicine called a Janus kinase (JAK) inhibitor. Xeljanz and Xeljanz XR is used to treat adults with moderately to severely active rheumatoid arthritis in whom methotrexate did not work well or cannot be tolerated.
Xeljanz and Xeljanz XR is used to treat adults with active psoriatic arthritis in which methotrexate or other similar medicines called nonbiologic disease-modifying antirheumatic drugs (DMARDs) did not work well or cannot be tolerated.
Xeljanz and Xeljanz XR is used to treat adults with moderately to severely active ulcerative colitis when medicines called tumor necrosis factor (TNF) blockers did not work well or cannot be tolerated.
Xeljanz and Xeljanz Oral Solution is used to treat patients 2 years of age and older with active polyarticular juvenile arthritis.
It is not known if Xeljanz and Xeljanz XR is safe and effective in people with Hepatitis B or C.
Xeljanz, Xeljanz XR and Xeljanz Oral Solution is not recommended for people with severe liver problems.
It is not known if Xeljanz and Xeljanz Oral Solution is safe and effective in children for treatment other than active polyarticular juvenile arthritis.
It is not known if Xeljanz XR is safe and effective in children.
Description
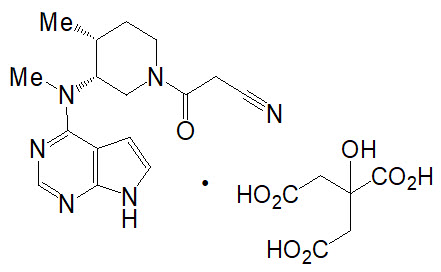
XELJANZ/XELJANZ XR (tofacitinib) tablets and XELJANZ (tofacitinib) Oral Solution are formulated with the citrate salt of tofacitinib, a JAK inhibitor.
Tofacitinib citrate is a white to off-white powder with the following chemical name: (3R,4R)-4-methyl-3-(methyl-7H-pyrrolo [2,3-d]pyrimidin-4-ylamino)-ß-oxo-1-piperidinepropanenitrile, 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
The solubility of tofacitinib citrate in water is 2.9 mg/mL.
Tofacitinib citrate has a molecular weight of 504.5 Daltons (or 312.4 Daltons as the tofacitinib free base) and a molecular formula of C16H20N6O∙C6H8O7. The chemical structure of tofacitinib citrate is:
XELJANZ is supplied for oral administration as a 5 mg white round, immediate-release film-coated tablet. Each tablet of XELJANZ contains 5 mg tofacitinib (equivalent to 8.08 mg tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
XELJANZ is supplied for oral administration as a 10 mg blue round, immediate-release film-coated tablet. Each 10 mg tablet of XELJANZ contains 10 mg tofacitinib (equivalent to 16.16 mg of tofacitinib citrate) and the following inactive ingredients: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
XELJANZ XR is supplied for oral administration as a 11 mg pink, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each 11 mg tablet of XELJANZ XR contains 11 mg tofacitinib (equivalent to 17.77 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide and triacetin. Printing ink contains, ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
XELJANZ XR is supplied for oral administration as a 22 mg beige, oval, extended-release film-coated tablet with a drilled hole at one end of the tablet band. Each 22 mg tablet of XELJANZ XR contains 22 mg tofacitinib (equivalent to 35.54 mg tofacitinib citrate) and the following inactive ingredients: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
XELJANZ Oral Solution is supplied for oral administration as a 1 mg/mL clear, colorless solution. Each 1 mL of XELJANZ Oral Solution contains 1 mg of tofacitinib (equivalent to 1.62 mg tofacitinib citrate) and the following inactive ingredients: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
Mechanism of Action
Tofacitinib is a Janus kinase (JAK) inhibitor. JAKs are intracellular enzymes which transmit signals arising from cytokine or growth factor-receptor interactions on the cellular membrane to influence cellular processes of hematopoiesis and immune cell function. Within the signaling pathway, JAKs phosphorylate and activate Signal Transducers and Activators of Transcription (STATs) which modulate intracellular activity including gene expression. Tofacitinib modulates the signaling pathway at the point of JAKs, preventing the phosphorylation and activation of STATs. JAK enzymes transmit cytokine signaling through pairing of JAKs (e.g., JAK1/JAK3, JAK1/JAK2, JAK1/TyK2, JAK2/JAK2). Tofacitinib inhibited the in vitro activities of JAK1/JAK2, JAK1/JAK3, and JAK2/JAK2 combinations with IC50 of 406, 56, and 1377 nM, respectively. However, the relevance of specific JAK combinations to therapeutic effectiveness is not known.
What is the most important information I should know about Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
Xeljanz, Xeljanz XR and Xeljanz Oral Solution may cause serious side effects including:
1. Serious infections. Xeljanz, Xeljanz XR and Xeljanz Oral Solution is a medicine that affects your immune system. Xeljanz, Xeljanz XR and Xeljanz Oral Solution can lower the ability of your immune system to fight infections. Some people can have serious infections while taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
- Your healthcare provider should test you for TB before starting Xeljanz, Xeljanz XR and Xeljanz Oral Solution and during treatment.
- Your healthcare provider should monitor you closely for signs and symptoms of TB infection during treatment with Xeljanz, Xeljanz XR and Xeljanz Oral Solution.
You should not start taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution if you have any kind of infection unless your healthcare provider tells you it is okay. You may be at a higher risk of developing shingles (herpes zoster). People taking the higher dose of Xeljanz (10 mg twice daily) or Xeljanz XR (22 mg one time each day) have a higher risk of serious infections and shingles.
Before starting Xeljanz, Xeljanz XR and Xeljanz Oral Solution, tell your healthcare provider if you:
- think you have an infection or have symptoms of an infection such as:
- fever, sweating, or chills
- muscle aches
- cough
- shortness of breath
- blood in phlegm
- weight loss
- warm, red, or painful skin or sores on your body
- diarrhea or stomach pain
- burning when you urinate or urinating more often than normal
- feeling very tired
- are being treated for an infection.
- get a lot of infections or have infections that keep coming back.
- have diabetes, chronic lung disease, HIV, or a weak immune system. People with these conditions have a higher chance for infections.
- have TB, or have been in close contact with someone with TB.
- live or have lived, or have traveled to certain parts of the country (such as the Ohio and Mississippi River valleys and the Southwest) where there is an increased chance for getting certain kinds of fungal infections (histoplasmosis, coccidioidomycosis, or blastomycosis). These infections may happen or become more severe if you use Xeljanz, Xeljanz XR and Xeljanz Oral Solution. Ask your healthcare provider if you do not know if you have lived in an area where these infections are common.
- have or have had hepatitis B or C.
After starting Xeljanz & Xeljanz XR, call your healthcare provider right away if you have any symptoms of an infection. Xeljanz & Xeljanz XR can make you more likely to get infections or make worse any infection that you have.
2. Increased risk of death in people 50 years of age and older with rheumatoid arthritis who have at least 1 heart disease (cardiovascular) risk factor and who are taking a higher than recommended dose of Xeljanz and Xeljanz XR. The recommended dose in patients with rheumatoid arthritis and psoriatic arthritis is Xeljanz 5 mg twice daily or Xeljanz XR 11 mg one time each day.
3. Cancer and immune system problems. Xeljanz, Xeljanz XR and Xeljanz Oral Solution may increase your risk of certain cancers by changing the way your immune system works.
- Lymphoma and other cancers including skin cancers can happen in patients taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution. People taking the higher dose of Xeljanz (10 mg twice daily) or Xeljanz XR (22 mg one time each day) have a higher risk of skin cancers. Tell your healthcare provider if you have ever had any type of cancer.
- Some people who have taken Xeljanz with certain other medicines to prevent kidney transplant rejection have had a problem with certain white blood cells growing out of control (Epstein Barr Virus-associated posttransplant lymphoproliferative disorder).
4. Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in patients with rheumatoid arthritis who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking a higher than recommended dose of Xeljanz and Xeljanz XR. The recommended dose in patients with rheumatoid arthritis and psoriatic arthritis is Xeljanz 5 mg twice daily or Xeljanz XR 11 mg one time each day. Blood clots in the lungs have also happened in patients with ulcerative colitis. Some people have died from these blood clots.
- Stop taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution and tell your healthcare provider right away if you develop signs and symptoms of a blood clot, such as sudden shortness of breath or difficulty breathing, chest pain, swelling of the leg or arm, leg pain or tenderness, or redness or discoloration in the leg or arm.
5. Tears (perforation) in the stomach or intestines.
- Tell your healthcare provider if you have had diverticulitis (inflammation in parts of the large intestine) or ulcers in your stomach or intestines. Some people taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution can get tears in their stomach or intestines. This happens most often in people who also take nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, or methotrexate. Tell your healthcare provider right away if you have fever and stomach-area pain that does not go away, and a change in your bowel habits.
6. Allergic reactions.
- Symptoms such as swelling of your lips, tongue, or throat, or hives (raised, red patches of skin that are often very itchy) that may mean you are having an allergic reaction have been seen in patients taking Xeljanz & Xeljanz XR. Some of these reactions were serious. If any of these symptoms occur while you are taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution, stop Xeljanz, Xeljanz XR and Xeljanz Oral Solution and call your healthcare provider right away.
7. Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start receiving Xeljanz, Xeljanz XR and Xeljanz Oral Solution and while you take Xeljanz, Xeljanz XR and Xeljanz Oral Solution to check for the following side effects:
- changes in lymphocyte counts. Lymphocytes are white blood cells that help the body fight off infections.
- low neutrophil counts. Neutrophils are white blood cells that help the body fight off infections.
- low red blood cell count. This may mean that you have anemia, which may make you feel weak and tired.
Your healthcare provider should routinely check certain liver tests.
You should not receive Xeljanz, Xeljanz XR and Xeljanz Oral Solution if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high.
Your healthcare provider may stop your Xeljanz, Xeljanz XR and Xeljanz Oral Solution treatment for a period of time if needed because of changes in these blood test results.
You may also have changes in other laboratory tests, such as your blood cholesterol levels. Your healthcare provider should do blood tests to check your cholesterol levels 4 to 8 weeks after you start receiving Xeljanz, Xeljanz XR and Xeljanz Oral Solution, and as needed after that. Normal cholesterol levels are important to good heart health.
See “What are the possible side effects of Xeljanz & Xeljanz XR?” for more information about side effects.
What should I tell my healthcare provider before taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
Before taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution, tell your healthcare provider about all of your medical conditions, including if you:
- have an infection. See “What is the most important information I should know about Xeljanz, Xeljanz XR and Xeljanz Oral Solution?”
- have had blood clots in the veins of your legs, arms, or lungs, or clots in the arteries in the past.
- have liver problems.
- have kidney problems.
- have any stomach area (abdominal) pain or been diagnosed with diverticulitis or ulcers in your stomach or intestines.
- have had a reaction to tofacitinib or any of the ingredients in Xeljanz, Xeljanz XR and Xeljanz Oral Solution.
- have recently received or are scheduled to receive a vaccine. People who take Xeljanz, Xeljanz XR and Xeljanz Oral Solution should not receive live vaccines. People taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution can receive non-live vaccines.
- plan to become pregnant or are pregnant. Xeljanz, Xeljanz XR and Xeljanz Oral Solution may affect the ability of females to get pregnant. It is not known if this will change after stopping Xeljanz, Xeljanz XR and Xeljanz Oral Solution. It is not known if Xeljanz, Xeljanz XR and Xeljanz Oral Solution will harm an unborn baby.
- Pregnancy Registry: Pfizer has a registry for pregnant women who take Xeljanz, Xeljanz XR and Xeljanz Oral Solution. The purpose of this registry is to check the health of the pregnant mother and her baby. If you are pregnant or become pregnant while taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution, talk to your healthcare provider about how you can join this pregnancy registry or you may contact the registry at 1-877-311- 8972 to enroll.
- plan to breastfeed or are breastfeeding. You and your healthcare provider should decide if you will take Xeljanz, Xeljanz XR and Xeljanz Oral Solution or breastfeed. You should not do both. After you stop your treatment with Xeljanz, Xeljanz XR and Xeljanz Oral Solution do not start breastfeeding again until:
- 18 hours after your last dose of Xeljanz or Xeljanz Oral Solution or
- 36 hours after your last dose of Xeljanz XR
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Xeljanz, Xeljanz XR and Xeljanz Oral Solution and other medicines may affect each other causing side effects.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Xeljanz, Xeljanz XR and Xeljanz Oral Solution and other medicines may affect each other causing side effects.
Especially tell your healthcare provider if you take:
- any other medicines to treat your rheumatoid arthritis, psoriatic arthritis, ulcerative colitis or polyarticular juvenile arthritis. You should not take tocilizumab (Actemra), etanercept (Enbrel), adalimumab (Humira), infliximab (Remicade), rituximab (Rituxan), abatacept (Orencia), anakinra (Kineret), certolizumab (Cimzia), golimumab (Simponi), ustekinumab (Stelara), secukinumab (Cosentyx), vedolizumab (Entyvio), azathioprine, cyclosporine, or other immunosuppressive drugs while you are taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution. Taking Xeljanz, Xeljanz XR and Xeljanz Oral Solution with these medicines may increase your risk of infection.
- medicines that affect the way certain liver enzymes work. Ask your healthcare provider if you are not sure if your medicine is one of these.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
- Take Xeljanz, Xeljanz XR and Xeljanz Oral Solution exactly as your healthcare provider tells you to take it. See the Xeljanz Oral Solution Instructions for use below.
- Take Xeljanz and Xeljanx Oral Solution 2 times a day with or without food.
- Take Xeljanz XR 1 time a day with or without food.
- Swallow Xeljanz XR tablets whole and intact. Do not crush, split, or chew.
- When you take Xeljanz XR, you may see something in your stool that looks like a tablet. This is the empty shell from the tablet after the medicine has been absorbed by your body.
- If you take too much Xeljanz, Xeljanz XR and Xeljanz Oral Solution, call your healthcare provider or go to the nearest hospital emergency room right away.
- For the treatment of psoriatic arthritis, take Xeljanz and Xeljanz XR in combination with methotrexate, sulfasalazine or leflunomide as instructed by your healthcare provider.
- Xeljanz XR should not be used instead of Xeljanz Oral Solution.
What are possible side effects of Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
Xeljanz, Xeljanz XR and Xeljanz Oral Solution may cause serious side effects, including:
- See “What is the most important information I should know about Xeljanz & Xeljanz XR?”
- Hepatitis B or C activation infection in people who carry the virus in their blood. If you are a carrier of the hepatitis B or C virus (viruses that affect the liver), the virus may become active while you use Xeljanz, Xeljanz XR and Xeljanz Oral Solution. Your healthcare provider may do blood tests before you start treatment with Xeljanz, Xeljanz XR and Xeljanz Oral Solution and while you are using Xeljanz, Xeljanz XR and Xeljanz Oral Solution. Tell your healthcare provider if you have any of the following symptoms of a possible hepatitis B or C infection:
- feel very tired o skin or eyes look yellow
- little or no appetite
- vomiting
- clay-colored bowel movements
- fevers
- chills
- stomach discomfort
- muscle aches
- dark urine
- skin rash
Common side effects of Xeljanz and Xeljanz XR in rheumatoid arthritis patients and psoriatic arthritis patients include:
- upper respiratory tract infections (common cold, sinus infections)
- headache
- diarrhea
- nasal congestion, sore throat, and runny nose (nasopharyngitis)
- high blood pressure (hypertension)
Common side effects of Xeljanz and Xeljanz XR in ulcerative colitis patients include:
- nasal congestion, sore throat, and runny nose (nasopharyngitis)
- increased cholesterol levels
- headache
- upper respiratory tract infections (common cold, sinus infections)
- increased muscle enzyme levels
- rash
- diarrhea
- shingles (herpes zoster)
Common side effects of Xeljanz and Xeljanz Oral Solution in polyarticular juvenile arthritis patients include:
- upper respiratory tract infections (common cold, sinus infections)
- nasal congestion, sore throat, and runny nose (nasopharyngitis)
- headache
- fever
- nausea
- vomiting
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Xeljanz, Xeljanz XR and Xeljanz Oral Solution. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Pfizer at 1-800-438-1985.
General information about the safe and effective use of Xeljanz, Xeljanz XR and Xeljanz Oral Solution
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Xeljanz, Xeljanz XR and Xeljanz Oral Solution for a condition for which it was not prescribed. Do not give Xeljanz, Xeljanz XR and Xeljanz Oral Solution to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about Xeljanz, Xeljanz XR and Xeljanz Oral Solution. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about Xeljanz, Xeljanz XR and Xeljanz Oral Solution that is written for health professionals.
How should I store Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
- Store Xeljanz and Xeljanz XR at room temperature between 68°F to 77°F (20°C to 25°C).
- Store Xeljanz Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C) in the original bottle and carton to protect from light.
- Safely throw away Xeljanz Oral Solution that is out of date or no longer needed. Use Xeljanz Oral Solution within 60 days of opening the bottle. Throw away (discard) remaining oral solution after 60 days.
Keep Xeljanz, Xeljanz XR and Xeljanz Oral Solution and all medicines out of the reach of children.
What are the ingredients in Xeljanz, Xeljanz XR and Xeljanz Oral Solution?
Active ingredient: tofacitinib citrate
Inactive ingredients:
Xeljanz 5mg: croscarmellose sodium, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
Xeljanz 10mg: croscarmellose sodium, FD&C Blue #1/Brilliant Blue FCF Aluminum Lake, FD&C Blue #2/Indigo Carmine Aluminum Lake, HPMC 2910/Hypromellose 6cP, lactose monohydrate, macrogol/PEG3350, magnesium stearate, microcrystalline cellulose, titanium dioxide, and triacetin.
Xeljanz XR 11mg: cellulose acetate, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, and triacetin. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron, propylene glycol, and shellac glaze.
Xeljanz XR 22mg: cellulose acetate, copovidone, FD&C Blue #2 Aluminum Lake, hydroxyethyl cellulose, hydroxypropyl cellulose, HPMC 2910/Hypromellose, magnesium stearate, red iron oxide, sorbitol, titanium dioxide, triacetin, and yellow iron oxide. Printing ink contains ammonium hydroxide, ferrosoferric oxide/black iron oxide, propylene glycol, and shellac glaze.
Xeljanz XR Oral Solution: grape flavor (natural), hydrochloric acid, lactic acid, purified water, sodium benzoate, sucralose, and xylitol.
Instructions for use for Xeljanz Oral Solution
Xeljanz (ZEL’ JANS’)
(tofacitinib)
Oral Solution
Important information about measuring Xeljanz Oral Solution:
Always use the oral dosing syringe that comes with Xeljanz Oral Solution to measure and take your prescribed dose. Ask your healthcare provider or pharmacist to show you how to measure your prescribed dose if you are not sure.
How should I store Xeljanz?
- Store Xeljanz Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C).
- Always store Xeljanz Oral Solution in the original bottle and carton to protect from light.
Keep Xeljanz and all medicines out of the reach of children.
Use Xeljanz Oral Solution within 60 days of opening the bottle. Throw away (discard) remaining Xeljanz Oral Solution after 60 days.
To help you remember when to throw away your bottle of Xeljanz Oral Solution, you can write the date when you first start to use it on the carton and below:
Date of first use ____ / ____ / ____.
Before each use:
Wash your hands with soap and water and place the items from the carton on a clean, flat surface.
Each carton of Xeljanz Oral Solution contains:
- 1 press-in bottle adapter
- 1 bottle of Xeljanz Oral Solution
- 1 oral dosing syringe
Step 1. Remove bottle from carton
Open the carton and remove the bottle of Xeljanz Oral Solution.
Step 2. Open bottle
Open the bottle by pushing down on the child-resistant cap and turning it to the left (counter-clockwise) as shown. Remove the seal off the top of the bottle (first time only).
Do not throw away the child-resistant cap.
Note: The bottle does not need to be shaken before use.
Step 3. Insert press-in bottle adapter (first time only)
Remove the press-in bottle adapter and oral dosing syringe from the plastic overwrap. With the bottle on a flat surface, push the ribbed end of the press-in bottle adapter all the way into the neck of the bottle with your thumbs while holding the bottle firmly.
Note: Do not remove the press-in bottle adapter from the bottle after it is inserted.
Step 4. Remove air from oral dosing syringe
Push the oral dosing syringe plunger all the way down to the tip of the syringe barrel to remove excess air.
Step 5. Insert the oral dosing syringe
Insert the oral dosing syringe tip into the upright bottle through the opening of the press-in bottle adapter until it is firmly in place.
Step 6. Withdraw dose from bottle
With the oral dosing syringe in place, turn the bottle upside down. Pull down on the plunger until the bottom of the plunger is even with the markings on the oral dosing syringe for your prescribed dose of oral solution.
If you see air bubbles in the oral dosing syringe, fully push the plunger in so that the oral solution flows back into the bottle. Then withdraw your prescribed dose of oral solution.
Step 7. Remove oral dosing syringe
Turn the bottle upright and place the bottle on a flat surface. Remove the oral dosing syringe from the press-in bottle adapter and bottle by pulling straight up on the oral dosing syringe barrel.
Step 8. Check the dose
Check that the correct dose was drawn up into the oral dosing syringe.
If the dose is not correct, insert the oral dosing syringe tip firmly into the press-in bottle adapter. Fully push in the plunger so that the oral solution flows back into the bottle. Repeat Step 6 and Step 7.
Step 9. Take the dose of Xeljanz
Place the tip of the oral dosing syringe into the inside of the cheek.
Slowly push the plunger all the way down to give all of the medicine in the oral dosing syringe. Make sure there is time to swallow the medicine.
Step 10. Close the bottle
Close the bottle tightly by turning the child-resistant cap to the right (clockwise), leaving the press-in bottle adapter in place.
Place the bottle back into the carton.
Close the carton to protect Xeljanz Oral Solution from light.
Step 11. Clean oral dosing syringe
Remove the plunger from the barrel by pulling the plunger and the barrel away from each other.
Rinse both with water after each use.
Allow to air dry. When the barrel and plunger are dry, put the oral dosing syringe back together by inserting the plunger into the barrel.
Store the oral dosing syringe with the Xeljanz Oral Solution.
Label
PRINCIPAL DISPLAY PANEL – 5 MG TABLET BOTTLE LABEL
- PROFESSIONAL SAMPLE-NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE - NDC 63539-012-02
- Pfizer
- Xeljanz™
(tofacitinib tablets) - 5 mg*
- 60 Tablets
Rx only
PRINCIPAL DISPLAY PANEL – 10 MG TABLET BOTTLE LABEL
- PROFESSIONAL SAMPLE – NOT FOR SALE
ALWAYS DISPENSE WITH MEDICATION GUIDE - NDC 63539-016-02
- Pfizer
- Xeljanz®
(tofacitinib) tablets - 10 mg*
- 10mg Is Recommended Only In
Ulcerative Colitis - 60 Tablets
Rx only

SRC: NLM .

