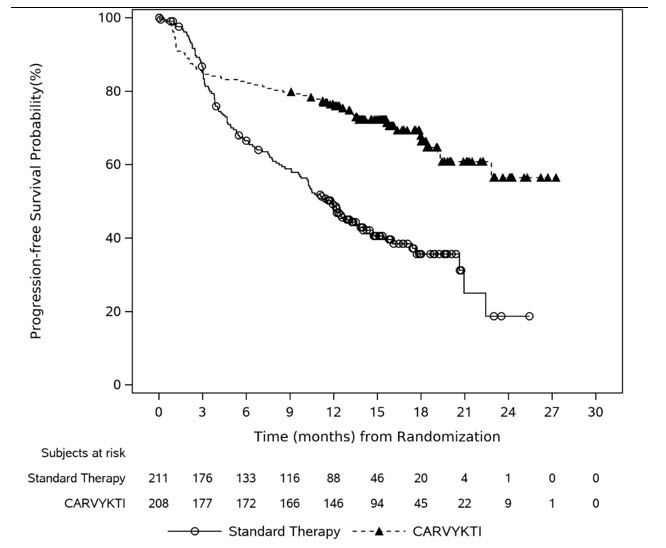
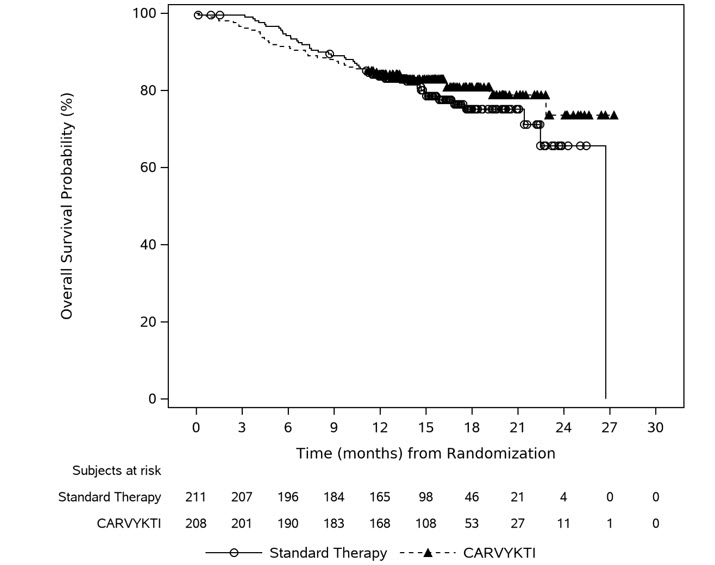
Carvykti
Generic Name: Ciltacabtagene autoleucel
Dosage form: Suspension for intravenous infusion
Medically reviewed by R Rash MD. Updated on 23 Mar 2022.
What exactly is Carvykti?
Carvykti is a genetically engineered autologous T cell immunotherapy that is directed by BCMA. Carvykti is synthesized from the patient’s mononuclear cells in the peripheral blood, which is collected by a conventional leukapheresis process. The mononuclear cells are enriched for T cells and genetically modified ex vivo using a replication-incompetent lentiviral vector to express a chimeric antigen receptor (CAR) with an anti-BCMA targeting domain made up of two single-domain antibodies linked to a 4-1BB costimulatory domain and a CD3-zeta signaling domain.
Anti-BCMA CAR T cells that have been transduced are grown in cell culture, washed, formed into a suspension, and cryopreserved. Before being shipped as a frozen suspension in a patient-specific infusion bag, the product must pass a sterility test. The anti-BCMA CAR T cells are frozen and injected back into the patient, where they may recognize and eradicate BCMA-expressing target cells.¶
Carvykti may contain Natural Killer (NK) cells in addition to T cells. Dimethyl sulfoxide (DMS) is present in the formulation at a concentration of 5%. (DMSO).
- Carvykti is a treatment to treat adult patients suffering from bone marrow cancer known as multiple myeloma. It is prescribed in cases where at least four other types of treatment have not been effective or have stopped working.
- Carvykti is a drug that is made by those white blood cells that you have which have been modified (genetically altered) to be able to recognize and attack the cells of your multiple myeloma.
Mechanisms of Action
- Carvykti is a BCMA-directed, genetically modified autologous T cell immunotherapy that includes reprogramming a patient’s own T cells with a transgene encoding a chimeric antigen receptor (CAR) that recognizes and kills BCMA-expressing cells. The Carvykti CAR protein has two BCMA-targeting single-domain antibodies with high avidity for human BCMA, as well as a 4-1BB co-stimulatory domain and a CD3-zeta (CD3-zeta) signaling cytoplasmic domain. The CAR enhances T cell activation, growth, and eradication of target cells after attaching to BCMA-expressing cells.
Important information
Carvykti could cause adverse reactions that are serious or life-threatening and could cause death. Consult your physician or seek emergency assistance right immediately if you experience any of the following symptoms:
- fever (100.4degF/38degC or more)
- Chills or shaking chills
- rapid or irregular heartbeat
- difficulties breathing
- Very low blood pressure
- dizziness/lightheadedness
- the effects on the nervous system. Some of them could occur just days or weeks after receiving the infusion. They may at first appear subtle, such as:
- experiencing confusion, feeling more disoriented, or less alert experiencing difficulty speaking or speech slurred, experiencing difficulty writing, reading, and being able to comprehend words, and memory loss
- loss of coordination in balance and movement, slower movements, and changes in handwriting
- personality changes, including a diminished capability to show emotion, less talkative, a lack of interest in pursuits, and decreased facial expression
- Tingling, numbness, and pain in feet and hands, trouble walking, arm or leg weakness, and trouble breathing
- Facial numbness, difficulty in moving facial muscles, and eyes
It is crucial to inform your healthcare provider that you’ve received Carvykti and also present them with your Carvykti Patient Card for Wallet. Your healthcare provider could give you additional medications to manage your symptoms.¶
Warnings and Precautions
- Recurrent and prolonged cytopenias: Patients could exhibit >=Grade 3 cytopenias after Carvykti infusion. Recurrences of Grade 3 or greater cytopenias can occur following the complete or partial recovery of cytopenias. Check blood counts prior to and following Carvykti infusion. Prolonged neutropenia can be linked to an increased chance of contracting an infection.
- Infections: Watch patients for symptoms and signs of infection and treat them appropriately.
- Hypogammaglobulinemia: Monitor and consider immunoglobulin replacement therapy.
- Hypersensitivity Reactions: Hypersensitivity reactions have happened. Check for hypersensitivity reactions while you are the infusion.
- Secondary Malignancies: In the case, the secondary malignancy is discovered following treatment with Carvykti you should contact Janssen Biotech, Inc. at 1-800-526-7736.
- Impacts on the Ability to Drive and use Machines Patients should not drive or engage in dangerous occupations or activities like operating heavy machinery that could be dangerous, for at least 8 weeks following having received Carvykti as well in case of a new neurologic toxicities onset.
Before you get Carvykti
Before receiving Carvykti, tell your doctor about your medical condition such as if you suffer from:
- Neurologic disorders that have occurred in the past or present (such as strokes, seizures, and new or worsening memory loss)
- Problems with breathing or the lungs
- Heart issues
- Liver issues
- Kidney issues
- An active or recent disease
- Low blood counts
What other drugs can impact Carvykti?
Inform your doctor about every medication you are taking, including prescription and over-the-counter medicines as well as vitamins or herbal remedies.
Certain products sold by commercial Human Immunodeficiency Virus (HIV) tests could incorrectly show an HIV-positive test result when you’re receiving treatment using Carvykti.
How can I receive Carvykti?
- Carvykti is made up of those white cells that you have, which means that your blood will be gathered via a procedure known as leukapheresis. The procedure could take up to six hours and might require repeating.
- The white blood cells go to a factory for the production of Carvykti. It will take about four to five weeks after the cells arrive at the manufacturing center and are ready to be returned to your doctor however, the timing could differ.
- When Carvykti is being developed, you could be offered other medications to treat multiple myeloma. This is to ensure that your myeloma does not worsen.
Prior to receiving Carvykti the doctor who treats you will prescribe chemotherapy for 3 days in order to prepare your body for the procedure.
Thirty-six to sixty minutes prior to the time you receive Carvykti You may receive additional medicines. They could include:
- remedies for allergic reactions (antihistamines)
- medicine to treat fever (such as Acetaminophen)
Once you are sure that your Carvykti is in good condition and your healthcare provider is satisfied, they will administer Carvykti to you via an infusion catheter (tube) that is inserted inside the vein (intravenous infusion). Your dose will be delivered via a single bag of infusion. Infusions typically last 30-60 minutes.
After receiving Carvykti You will be monitored by the healthcare facility that is certified by the FDA in which you received the treatment for a minimum of 10 days following the infusion.
You must plan to stay near the place in which you received your treatment for a minimum of 4 weeks. Your doctor will be able to make sure that your treatment is working and will assist you to deal with any adverse reactions that might happen.
It is possible to be admitted to the hospital when you experience dangerous side effects. This will continue until your symptoms are controlled and safe to quit the hospital.
Your physician will be able to conduct blood tests in order to monitor your improvement. It is crucial to test your blood. If you don’t make an appointment, you should contact your healthcare professional as soon as you can to make a new appointment.
Dosing information
The usual adult dose for Multiple Myeloma
Recommended dose range is 0.5-1.0x106CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×108 CAR-positive viable T cells per single-dose infusion.
For autologous use only. For intravenous use only.
- Administer a lymphodepleting regimen of cyclophosphamide and fludarabine before infusion of Carvykti.
- Do NOT use a leukodepleting filter.
- Verify the patient’s identity prior to infusion.
- Premedicate with acetaminophen and an H1-antihistamine.
- Avoid prophylactic use of systemic corticosteroids.
- Confirm availability of tocilizumab prior to infusion.
- Dosing of Carvykti is based on the number of chimeric antigen receptor (CAR)-positive viable T cells.
- Recommended dose range is 0.5–1.0×106 CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×108 CAR-positive viable T cells per single-dose infusion.
- Administer Carvykti at a REMS-certified healthcare facility
What should I be aware of following the initiation of Carvykti?
- Avoid driving, using heavy machinery, or engaging in other activities which could pose danger when you’re not alert for at least 8 weeks following the time you receive Carvykti. The reason for this is that the treatment could result in memory and coordination issues insomnia, sleepiness, confusion seizures, dizziness, or other neurologic effects explained by your doctor.
- You should not receive certain live vaccines, also known as live vaccines, prior to and following Carvykti treatment. Discuss with your healthcare professional for advice on any vaccinations.
- Don’t donate organs, blood, tissue, or cells for transplantation.
Detailed Carvykti dosage information
Carvykti side effects
The most frequent adverse effects are:
- fever (100.4degF/38degC or more) chills, fever
- dizziness or lightheadedness
- headache muscles or joint pain, feeling extremely tired
- Affected mental state, disorientation
- Infections
- chest pain
- very low concentrations of antibodies (immunoglobulins) in blood
- cough, feeling short of breath
- nausea, diarrhea loss of appetite, and constipation
- rapid or irregular heartbeat
- issues with blood issues with blood clotting
Carvykti can trigger a frequent side effect, a condition known as CRS, or cytokine release syndrome, and can be very serious or even fatal. The symptoms of CRS are the following: fever, breathing difficulties dizziness or lightheadedness headache, nausea, fast heartbeat or blood pressure that is low, or fatigue. Contact your healthcare professional immediately if you experience fever or experience any of these other symptoms following the administration of Carvykti.
Carvykti increases the risk of developing life-threatening infections that could lead to death. Contact your healthcare professional immediately if you experience symptoms of chills, fever, or any indications or signs of infection.
Carvykti may cause a variety of neurologic effects, and some could be fatal or severe.
The symptoms include but aren’t included in confusion and disorientation, loss of awareness, seizure, trouble talking, reading, or writing as well as tremors, slow movement or changes in personality seizures as well as depression, tingling, and the feeling of numbness in feet and hands or arm weakness, as well as facial Numbness.
Carvykti may decrease one or more blood cells (red blood cells white blood cells or platelets (cells that assist in causing blood clots) This can cause you to feel tired or weak or increase the risk of bleeding or serious infection. Following treatment, your doctor will analyze your blood for this. Inform your healthcare provider immediately if you notice an illness that causes chills, fever, or any indications or signs of infection, are experiencing fatigue, or are suffering from bleeding or bruising.
Being a carrier of Carvykti present in your blood can result in some medical Human Immunodeficiency Virus (HIV) tests to falsely show an HIV positive result, even though you could be HIV-negative.
There are many possible adverse consequences. Consult your doctor for any adverse symptoms. You can report any side effects to the FDA via 800-FDA-1088.
General information on the safe and efficient use of Carvykti.
Some medicines are used for reasons other than those listed in the Medication Guide. If you’d like more details, consult your doctor. You could ask your doctor to provide you with information that has been designed for health professionals. For more information, visit www.Carvykti.com or contact 1-800-526-7736.
Patients counseling information
Encourage the patient to study the FDA-approved labeling for patients (Medication Guidance). Be sure to inform patients about the possibility of failure in manufacturing (around 18% (17/97 in the study)(in the clinical study). If there is the manufacturing process fails and the second production of this medication could be tried. Additionally, when the patient is waiting for the drug, further anticancer treatments (other than lymphodepletion) could be required and can increase the chance of adverse reactions in the period prior to infusion which may hinder or stop administering Carvykti.
Instruct the patients to be observed on a daily basis for the first 10 consecutive days after the infusion in a REMS-certified healthcare facility. Also, advise patients to stay within the vicinity of a health facility for at least 4 weeks after the infusion.
Before infusion, inform your patients about the following dangers and urge them to seek medical attention in case of any of these warning signs and symptoms
Cytokine Release Syndrome (CRS)
Signs or symptoms of CRS, including fever, chills, fatigue, headache, tachycardia, hypotension, hypoxia, dizziness/lightheadedness, or organ toxicities
Neurologic Toxicities
Affects or signs that can be related to neurologic processes Some of them occur in the days, weeks or even months after the infusion, including
- ICANS e.g. Aphasia or encephalopathy decreased levels of awareness, seizure dysgraphia, delirium
- The signs of Parkinsonism include e.g. tremor micrographia or bradykinesia stiffness, shuffling gait tilted posture, masked facial features, apathy, flat affect sleepiness, lethargy
- Guillain Barre Syndrome: e.g., motor weakness, polyradiculoneuritis
- The peripheral neuropathy e.g. peripheral sensory or motor nerve problems
- Cranial Nerve Palsies: e.g., facial paralysis, facial numbness
Recurrent and Protracted Cytopenias
The signs or symptoms that are related to bone marrow suppression such as thrombocytopenia, neutropenia, anemia, or febrile neutropenia last for many weeks or months. The symptoms or signs associated with bone marrow deficiency can be recurrent.
Infections
Aspects or signs that suggest the infection
Hypersensitivity Reactions
The signs or symptoms that are associated with hypersensitivity reactions include chest tightness, flushing the tachycardia, and breathing difficulties
Instruct patients on the necessity to:
- Monitor blood counts prior to and following Carvykti infusion
- Call Janssen Biotech, Inc. by calling 1-800-526-7736 in the event that they are diagnosed with secondary malignancy
- Avoid driving or engaging in dangerous occupations or activities such as operating dangerous or heavy equipment for at least 8 weeks following treatment, as well in case of new symptoms of neurologic toxicities.
- Inform their doctor about the treatment they received with Carvykti prior to receiving a live virus vaccination
Label
70 ml label
- Ciltacabtagene autoleucel
CARVYKTI - Suspension for Intravenous Infusion
- Dose: One sterile bag for infusion.
- Storage: Store and transport in a vapor phase of
liquid nitrogen ≤ -120°C (-184°F). Thaw before using. - Rx only
30 ml Label
- Ciltacabtagene autoleucel
- CARVYKTI™
- Suspension for Intravenous Infusion
- Dose: One sterile bag for infusion.
- Rx only
Additional details
Always consult your doctor to make sure the information presented on this page is applicable to your particular situation.
