Triumeq
Generic name: abacavir, dolutegravir, and lamivudine
Brand names: Triumeq
Medically reviewed by A Ras MD. Last updated April 7, 2022
Important information
TRIUMEQ and TRIUMEQ PD can cause serious side effects, including:
- Serious allergic reactions (hypersensitivity reaction) that can cause death have happened with TRIUMEQ or TRIUMEQ PD and other abacavir-containing products. Your risk of this allergic reaction to abacavir is much higher if you have a gene variation called HLA-B*5701. Your healthcare provider can determine with a blood test if you have this gene variation.
If you get a symptom from 2 or more of the following groups while taking TRIUMEQ or TRIUMEQ PD, call your healthcare provider right away to find out if you should stop taking TRIUMEQ or TRIUMEQ PD.
| Symptom(s) | |
| Group 1 | Fever |
| Group 2 | Rash |
| Group 3 | Nausea, vomiting, diarrhea, abdominal (stomach area) pain |
| Group 4 | Generally ill feeling, extreme tiredness, or achiness |
| Group 5 | Shortness of breath, cough, sore throat |
A list of these symptoms is on the Warning Card your pharmacist gives you. Carry this Warning Card with you at all times.
If you stop TRIUMEQ or TRIUMEQ PD because of an allergic reaction, never take TRIUMEQ, TRIUMEQ PD (abacavir, dolutegravir and lamivudine), or any other medicine that contains abacavir or dolutegravir (DOVATO, EPZICOM, JULUCA, TIVICAY, TIVICAY PD, TRIZIVIR, or ZIAGEN) again.
- If you have an allergic reaction, dispose of any unused TRIUMEQ or TRIUMEQ PD. Ask your pharmacist how to properly dispose of medicines.
- If you take TRIUMEQ, TRIUMEQ PD or any other abacavir-containing medicine again after you have had an allergic reaction, within hours you may get life-threatening symptoms that may include very low blood pressure or
If you stop TRIUMEQ or TRIUMEQ PD for any other reason, even for a few days, and you are not allergic to TRIUMEQ or TRIUMEQ PD, talk with your healthcare provider before taking it again. Taking TRIUMEQ or TRIUMEQ PD again can cause a serious allergic or life-threatening reaction, even if you never had an allergic reaction to it before.
If your healthcare provider tells you that you can take TRIUMEQ or TRIUMEQ PD again, start taking it when you are around medical help or people who can call a healthcare provider if you need one.
- Worsening of Hepatitis B virus (HBV) infection. Your healthcare provider will test you for HBV infection before you start treatment with TRIUMEQ or TRIUMEQ PD. If you have HBV infection and take TRIUMEQ or TRIUMEQ PD, your HBV may get worse (flare-up) if you stop taking TRIUMEQ or TRIUMEQ PD. A “flare-up” is when your HBV infection suddenly returns in a worse way than before.
- Do not run out of TRIUMEQ or TRIUMEQ PD. Refill your prescription or talk to your healthcare provider before your TRIUMEQ or TRIUMEQ PD is all gone.
- Do not stop TRIUMEQ or TRIUMEQ PD without first talking to your healthcare provider.
- If you stop taking TRIUMEQ or TRIUMEQ PD, your healthcare provider will need to check your health often and do blood tests regularly for several months to check your liver function and monitor your HBV infection. It may be necessary to give you a medicine to treat hepatitis B. Tell your healthcare provider about any new or unusual symptoms you may have after you stop taking TRIUMEQ or TRIUMEQ PD.
- Resistant HBV. If you have human immunodeficiency virus-1 (HIV-1) and HBV, the HBV can change (mutate) during your treatment with TRIUMEQ or TRIUMEQ PD and become harder to treat (resistant). Your healthcare provider may give you other medicines to treat HBV infection if you have HIV-1 and HBV infections and take TRIUMEQ or TRIUMEQ PD.
For more information about side effects, see “What are the possible side effects of TRIUMEQ or TRIUMEQ PD?”
What is TRIUMEQ and TRIUMEQ PD?
TRIUMEQ and TRIUMEQ PD are prescription medicines used to treat HIV-1 infection in adults and children who weigh at least 22 pounds (10 kg).
HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS).
TRIUMEQ and TRIUMEQ PD contain the prescription medicines abacavir, dolutegravir, and lamivudine.
- TRIUMEQ or TRIUMEQ PD should not be used by itself in people who have resistance to certain types of medicines.
It is not known if TRIUMEQ PD is safe and effective in children who weigh less than 22 pounds (10 kg).
Do not take TRIUMEQ or TRIUMEQ PD if you:
- have a certain type of gene variation called the HLA-B*5701 allele. Your healthcare provider will test you for this before prescribing treatment with TRIUMEQ or TRIUMEQ PD.
- are allergic to abacavir, dolutegravir, lamivudine, or any of the ingredients in TRIUMEQ or
TRIUMEQ PD. See the end of this Medication Guide for a complete list of ingredients in TRIUMEQ and TRIUMEQ PD.
- take dofetilide. Taking TRIUMEQ or TRIUMEQ PD and dofetilide can cause side effects that may be serious or life-threatening.
- have certain liver problems.
Before you take TRIUMEQ or TRIUMEQ PD
tell your healthcare provider about all of your medical conditions, including if you:
- have been tested and know whether or not you have a particular gene variation called HLA-B*5701.
- have or have had liver problems, including hepatitis B or C virus infection.
- have kidney problems.
- have heart problems, smoke, or have diseases that increase your risk of heart disease such as high blood pressure, high cholesterol, or diabetes.
- drink alcohol or take medicines that contain alcohol.
- are pregnant or plan to become pregnant. One of the medicines in TRIUMEQ and TRIUMEQ PD called dolutegravir may harm your unborn baby.
- Your healthcare provider may prescribe a different medicine than TRIUMEQ if you are planning to become pregnant or if pregnancy is confirmed during the first 12 weeks of pregnancy.
- If you can become pregnant, your healthcare provider may perform a pregnancy test before you start treatment with TRIUMEQ.
- If you can become pregnant, you and your healthcare provider should talk about the use of effective birth control (contraception) during treatment with TRIUMEQ. o Tell your healthcare provider right away if you are planning to become pregnant, you become pregnant, or think you may be pregnant during treatment with TRIUMEQ.
Pregnancy Registry. There is a pregnancy registry for individuals who take TRIUMEQ and TRIUMEQ PD during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your healthcare provider about how you can take part in this registry.
- are breastfeeding or plan to breastfeed. Do not breastfeed if you take TRIUMEQ.
- You should not breastfeed if you have HIV-1 because of the risk of passing HIV-1 to your baby.
- Two of the medicines in TRIUMEQ and TRIUMEQ PD (abacavir and lamivudine) pass into your breastmilk.
Tell your healthcare provider about all the medicines you take, including prescription and over-thecounter medicines, vitamins, and herbal supplements.
Some medicines interact with TRIUMEQ or TRIUMEQ PD. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine.
You can ask your healthcare provider or pharmacist for a list of medicines that interact with TRIUMEQ or TRIUMEQ PD.
Do not start taking a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take TRIUMEQ or TRIUMEQ PD with other medicines.¶
How should I take TRIUMEQ or TRIUMEQ PD?
Read the Instructions for Use at the end of this Medication Guide for detailed instructions on how to prepare a dose of TRIUMEQ PD tablets for oral suspension.
- Take TRIUMEQ or TRIUMEQ PD exactly as your healthcare provider tells you to take it.
- Do not change your dose, switch medicines or stop taking TRIUMEQ or TRIUMEQ PD without talking with your healthcare provider first.
- TRIUMEQ tablets are not the same as TRIUMEQ PD tablets for oral suspension and cannot be substituted for each other. Check to make sure you receive the correct dosage form each time you or your child’s prescription is filled to avoid using the wrong medicine.
- Your child’s healthcare provider will prescribe TRIUMEQ or TRIUMEQ PD based on your child’s weight.
- TRIUMEQ PD tablets for oral suspension should be dispersed in drinking water.
- Do not chew, cut, or crush TRIUMEQ tablets or TRIUMEQ PD tablets for oral suspension.
- TRIUMEQ or TRIUMEQ PD may be taken with or without food.
- If you take antacids, laxatives, or other medicines that contain aluminum, magnesium, or buffered medicines, TRIUMEQ or TRIUMEQ PD should be taken at least 2 hours before or 6 hours after you take these medicines.
- If you need to take iron or calcium supplements by mouth during treatment with TRIUMEQ or TRIUMEQ PD:
- If you takeTRIUMEQ or TRIUMEQ PD with food, you may take these supplements at the same time that you take TRIUMEQ or TRIUMEQ PD.
- If you do not take TRIUMEQ or TRIUMEQ PD with food, take TRIUMEQ or TRIUMEQ PD at least 2 hours before or 6 hours after you take these supplements.
- If you miss a dose of TRIUMEQ or TRIUMEQ PD, take it as soon as you remember. Do not take 2 doses at the same time or take more than your healthcare provider tells you to take.
- Stay under the care of a healthcare provider during treatment with TRIUMEQ or TRIUMEQ PD.
- Do not run out of TRIUMEQ or TRIUMEQ PD. The virus in your blood may increase and the virus may become harder to treat. When your supply starts to run low, get more from your healthcare provider or pharmacy.
If you take too much TRIUMEQ or TRIUMEQ PD, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of TRIUMEQ or TRIUMEQ PD?
TRIUMEQ or TRIUMEQ PD can cause serious side effects, including
- Liver problems. People with a history of hepatitis B or C virus may have an increased risk of developing new or worsening changes in certain liver function tests during treatment with TRIUMEQ or TRIUMEQ PD. Liver problems including liver failure have also happened with TRIUMEQ or TRIUMEQ PD in people without a history of liver disease or other risk factors. Liver failure resulting in liver transplant has also been reported with TRIUMEQ. Your healthcare provider may do blood tests to check your liver. Call your healthcare provider right away if you develop any of the signs or symptoms of liver problems listed below.
- dark or “tea-colored” urine o pain, aching, or tenderness on the right side o light colored stools (bowel movements) of your stomach area
- Too much lactic acid in your blood (lactic acidosis). Too much lactic acid is a serious medical emergency that can lead to death. Call your healthcare provider right away if you get any of the following symptoms that could be signs of lactic acidosis:
- feel very weak or tired o feel cold, especially in your arms and legs o unusual (not normal) muscle pain o feel dizzy or lightheaded o trouble breathing o have a fast or irregular heartbeat
- stomach pain with nausea and vomiting
- Lactic acidosis can also lead to severe liver problems, which can lead to death. Your liver may become large (hepatomegaly) and you may develop fat in your liver (steatosis). Call your healthcare provider right away if you get any of the signs or symptoms of liver problems which are listed above under “Liver problems”.
- You may be more likely to get lactic acidosis or severe liver problems if you are female or very overweight (obese).
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having new symptoms after you start taking TRIUMEQ or TRIUMEQ PD.
- Heart attack. Some HIV-1 medicines including TRIUMEQ or TRIUMEQ PD may increase your risk of heart attack.
- The most common side effects of TRIUMEQ include:
- trouble sleeping
- headache
- tiredness
These are not all the possible side effects of TRIUMEQ or TRIUMEQ PD.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store TRIUMEQ or TRIUMEQ PD?
- Store TRIUMEQ and TRIUMEQ PD at room temperature between 68°F to 77°F (20°C to 25°C) in the original bottle. Keep the bottle tightly closed and protect it from moisture.
- The bottle of TRIUMEQ and TRIUMEQ PD contains a desiccant packet to help keep your medicine dry (protect it from moisture). Do not remove the desiccant packet from the bottle.
General information about the safety
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TRIUMEQ or TRIUMEQ PD for a condition for which it was not prescribed. Do not give TRIUMEQ or TRIUMEQ PD to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TRIUMEQ or TRIUMEQ PD that is written for health professionals.
What are the ingredients in TRIUMEQ and TRIUMEQ PD?
Active ingredients: abacavir, dolutegravir, and lamivudine Inactive ingredients:
TRIUMEQ tablets: D-mannitol, magnesium stearate, microcrystalline cellulose, povidone, and sodium starch glycolate. Tablet film-coating contains: iron oxide black, iron oxide red, macrogol/PEG, polyvinyl alcohol–part hydrolyzed, talc, and titanium oxide.
TRIUMEQ PD tablets for oral suspension: acesulfame potassium, crospovidone, mannitol, microcrystalline cellulose, povidone K29/32, silicified microcrystalline cellulose, sodium starch glycolate, sodium stearyl fumarate, strawberry cream flavor, and sucralose.
Tablet film-coating contains: ferric oxide yellow, macrogol/PEG, polyvinyl alcohol-part hydrolyzed, talc, and titanium dioxide.
INSTRUCTIONS FOR USE
TRIUMEQ PD (abacavir, dolutegravir, and lamivudine) tablets for oral suspension
This Instructions for Use contains information on how to take TRIUMEQ PD.
Read this Instructions for Use before your child starts taking TRIUMEQ PD for the first time and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your child’s medical condition or treatment. Talk to your healthcare provider or pharmacist if you have any questions on how to prepare or give the prescribed dose of TRIUMEQ PD
Important information
- Give TRIUMEQ PD exactly as your healthcare provider tells you.
- Each time you receive your child’s prescription, check the bottle to make sure that you received the correct dosage form of TRIUMEQ PD. Contact your pharmacist or healthcare provider if you did not receive the correct dosage form.
- Do not chew, cut, or crush the tablets for oral suspension.
- If you forget to give a dose of TRIUMEQ PD, give it as soon as you remember. Do not give 2 doses at the same time or give more than your healthcare provider has prescribed.
- If your child does not or cannot take the full dose, call your healthcare provider.
- If you give too much TRIUMEQ PD, get emergency medical help right away.
For more information about TRIUMEQ PD, read the Medication Guide.
| Your pack contains: | |
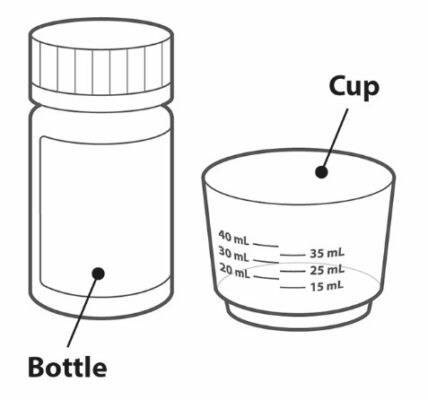 |
• A bottle containing 90 TRIUMEQ PD tablets for oral suspension
• Dosing cup |
| You will also need: • Clean drinking water. | |
| Getting Ready
Step 1. Pour water |
|

Figure A |
• Pour 20 mL of clean drinking water into the cup.
See Figure A. Use drinking water only. Do not use any other drink or food to prepare the dose. |
| Step 2. Prepare the medicine | |
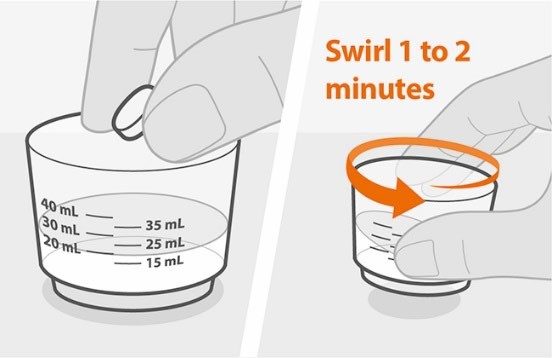
Figure B Figure C |
• Add the prescribed number of tablet(s) to the water. See Figure B.
• Swirl the cup gently for 1 to 2 minutes to disperse the tablet(s). The medicine will become cloudy. Take care not to spill any of the medicine. See Figure C. • Check that the medicine is ready. If there are any lumps of tablet, swirl the cup until they are gone. |
| If you spill any medicine, clean up the spill.
Throw away the rest of the prepared medicine and |
make a new dose. |
| You must give the dose of medicine within 30 m | inutes of preparing the dose. If it has been |
| more than 30 minutes, wash away all the dose in th medicine. | e cup using water and prepare a new dose of |
| Giving the medicine
Step 3. Give the medicine |
|
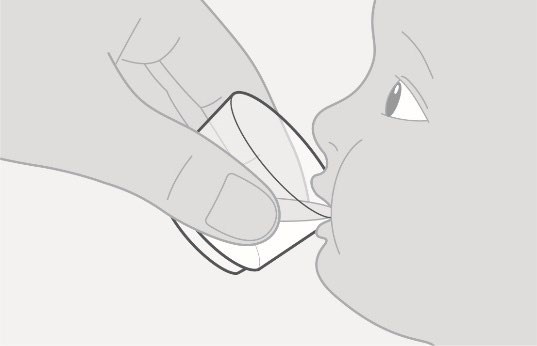
Figure D |
• Make sure that the child is upright. Give all the prepared medicine to the child. See Figure D.
• Add another 15 mL of drinking water to the cup, swirl, and give it all to the child. • Repeat if any medicine remains in the cup to make sure the child gets the full dose. |
| Clean
Step 4. Clean the dosing cup |
|
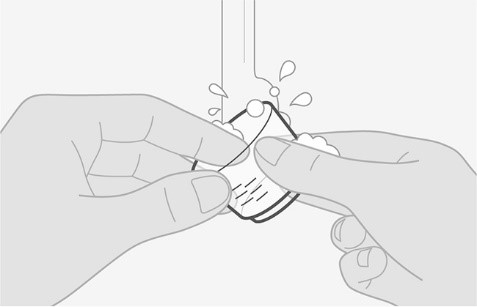
Figure E |
• Wash the cup with water. See Figure E.
• The cup will need to be clean before preparing the next dose. |
| Storage Information
• Store TRIUMEQ PD tablets for oral suspension at room temperature between 68°F to 77°F (20°C to 25°C) in the original bottle. Keep the bottle tightly closed and protect from moisture. • The TRIUMEQ PD bottle contains a desiccant packet to help keep your medicine dry (protect it from moisture). Do not remove the desiccant packet from the bottle. Keep TRIUMEQ PD and all medicines out of the reach of children. |
|
| Disposal Information
When all the tablets in the bottle have been taken or are no longer needed, throw away the bottle and cup. Dispose of them using your local household waste guidelines. You will get a new cup in your next pack. |
|
