Qudexy XR
Generic name: topiramate
Brand names: Eprontia, Qudexy XR Sprinkle, Topamax, Topamax Sprinkle, Trokendi XR
Drug class: Carbonic anhydrase inhibitor anticonvulsants
Medically reviewed by A Ras MD.
What is Qudexy XR?
Qudexy XR is a prescription medicine used to treat certain types of seizures (partial-onset seizures and primary generalized tonic-clonic seizures) in adults and children 2 years of age and older. It is also used with other medicines to treat certain types of seizures (partial-onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome) in adults and children 2 years of age and older.
Qudexy XR is used to prevent migraine headaches in adults and adolescents 12 years of age and older.
Description
Topiramate, USP, is a sulfamate-substituted monosaccharide. QUDEXY XR (topiramate) extended-release capsules are available as 25 mg, 50 mg, 100 mg, 150 mg, and 200 mg capsules for oral administration as whole capsules or opened and sprinkled onto a spoonful of soft food.
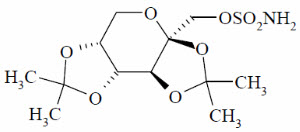
Topiramate is a white to off-white powder. Topiramate is freely soluble in polar organic solvents such as acetonitrile and acetone; and very slightly soluble to practically insoluble in non-polar organic solvents such as hexanes. Topiramate has the molecular formula C12H21NO8S and a molecular weight of 339.4. Topiramate is designated chemically as 2,3:4,5-Di-O-isopropylidene-β-D-fructopyranose sulfamate and has the following structural formula:
QUDEXY XR (topiramate) extended-release capsules contain beads of topiramate in a capsule. The inactive ingredients are microcrystalline cellulose, hypromellose 2910, ethylcellulose, diethyl phthalate.
In addition, the capsule shells for all strengths contain hypromellose 2910, titanium dioxide, black iron oxide, red iron oxide and/or yellow iron oxide, black pharmaceutical ink, and white pharmaceutical ink (200 mg only).
Mechanism of Action
The precise mechanisms by which topiramate exerts its anticonvulsant and preventive migraine effects are unknown; however, preclinical studies have revealed four properties that may contribute to topiramate’s efficacy for epilepsy and the preventive treatment of migraine. Electrophysiological and biochemical evidence suggests that topiramate, at pharmacologically relevant concentrations, blocks voltage-dependent sodium channels, augments the activity of the neurotransmitter gamma-aminobutyrate at some subtypes of the GABA-A receptor, antagonizes the AMPA/kainate subtype of the glutamate receptor, and inhibits the carbonic anhydrase enzyme, particularly isozymes II and IV.
What is the most important information I should know about Qudexy XR?
Qudexy XR may cause eye problems. Serious eye problems include:
- any sudden decrease in vision with or without eye pain and redness,
- a blockage of fluid in the eye causing increased pressure in the eye (secondary angle closure glaucoma).
These eye problems can lead to permanent loss of vision if not treated. You should call your healthcare provider right away if you have any new eye symptoms, including any new problems with your vision.
Qudexy XR may cause decreased sweating and increased body temperature (fever). People, especially children, should be watched for signs of decreased sweating and fever, especially in hot temperatures. Some people may need to be hospitalized for this condition. If you have a high fever, a fever that does not go away, or decreased sweating develops, call your healthcare provider right away.
Qudexy XR can increase the level of acid in your blood (metabolic acidosis). If left untreated, metabolic acidosis can cause brittle or soft bones (osteoporosis, osteomalacia, osteopenia), kidney stones, can slow the rate of growth in children, and may possibly harm your baby if you are pregnant. Metabolic acidosis can happen with or without symptoms. Sometimes people with metabolic acidosis will:
- feel tired
- not feel hungry (loss of appetite)
- feel changes in heartbeat
- have trouble thinking clearly
Your healthcare provider should do a blood test to measure the level of acid in your blood before and during your treatment with Qudexy XR.
If you are pregnant, you should talk to your healthcare provider about whether you have metabolic acidosis.
Like other antiepileptic drugs, Qudexy XR may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
Do not stop Qudexy XR without first talking to a healthcare provider.
- Stopping Qudexy XR suddenly can cause serious problems.
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
How can I watch for early symptoms of suicidal thoughts and actions?
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
Qudexy XR can harm your unborn baby.
- If you take Qudexy XR during pregnancy, your baby has a higher risk for birth defects called cleft lip and cleft palate. These defects can begin early in pregnancy, even before you know you are pregnant.
- Cleft lip and cleft palate may happen even in children born to women who are not taking any medicines and do not have other risk factors.
- There may be other medicines to treat your condition that have a lower chance of birth defects.
- All women of childbearing age should talk to their healthcare providers about using other possible treatments instead of Qudexy XR. If the decision is made to use Qudexy XR, you should use effective birth control (contraception) unless you are planning to become pregnant. You should talk to your healthcare provider about the best kind of birth control to use while you are taking Qudexy XR.
- Tell your healthcare provider right away if you become pregnant while taking Qudexy XR. You and your healthcare provider should decide if you will continue to take Qudexy XR while you are pregnant.
- If you take Qudexy XR during pregnancy, your baby may be smaller than expected at birth. The long-term effects of this are not known. Talk to your healthcare provider if you have any questions about this risk during pregnancy.
- Metabolic acidosis may have harmful effects on your baby. Talk to your healthcare provider if Qudexy XR has caused metabolic acidosis during your pregnancy.
- Pregnancy Registry: If you become pregnant while taking Qudexy XR, talk to your healthcare provider about registering with the North American Antiepileptic Drug Pregnancy Registry. You can enroll in this registry by calling 1-888-233-2334. The purpose of this registry is to collect information about the safety of Qudexy XR and other antiepileptic drugs during pregnancy.
What should I tell my healthcare provider before taking Qudexy XR?
Before taking Qudexy XR, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had depression, mood problems or suicidal thoughts or behavior
- have kidney problems, kidney stones or are getting kidney dialysis
- have a history of metabolic acidosis (too much acid in the blood)
- have liver problems
- have weak, brittle or soft bones (osteomalacia, osteoporosis, osteopenia, or decreased bone density)
- have lung or breathing problems
- have eye problems, especially glaucoma
- have diarrhea
- have a growth problem
- are on a diet high in fat and low in carbohydrates, which is called a ketogenic diet
- are having surgery
- are pregnant or plan to become pregnant
- are breastfeeding or plan to breastfeed. Qudexy XR passes into breast milk. Breastfed babies may be sleepy or have diarrhea. It is not known if the Qudexy XR that passes into breast milk can cause other serious harm to your baby. Talk to your healthcare provider about the best way to feed your baby if you take Qudexy XR.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Qudexy XR and other medicines may affect each other causing side effects.
Especially tell your healthcare provider if you take:
- Valproic acid (such as Depakene or Depakote)
- any medicines that impair or decrease your thinking, concentration, or muscle coordination
- birth control pills. Qudexy XR may make your birth control pills less effective. Tell your healthcare provider if your menstrual bleeding changes while you are taking birth control pills and Qudexy XR.
Ask your healthcare provider if you are not sure if your medicine is listed above.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. Do not start a new medicine without talking with your healthcare provider.
How should I take Qudexy XR?
- Take Qudexy XR exactly as your healthcare provider tells you to.
- Your healthcare provider may change your dose. Do not change your dose without talking to your healthcare provider.
- Qudexy XR capsules may be swallowed whole or, if you cannot swallow the capsule whole, you may carefully open the Qudexy XR capsule and sprinkle the medicine on a spoonful of soft food like applesauce.
- Swallow the food and medicine mixture right away. Do not store the food and medicine mixture to use later.
- Do not crush or chew Qudexy XR before swallowing.
- Drink plenty fluids during the day. This may help prevent kidney stones while taking Qudexy XR.
- If you take too much Qudexy XR, call your healthcare provider right away or go to the nearest emergency room.
- Qudexy XR can be taken before, during, or after a meal.
- If you miss a single dose of Qudexy XR, take it as soon as you can. If you have missed more than one dose, you should call your healthcare provider for advice.
- Do not stop taking Qudexy XR without talking to your healthcare provider. Stopping Qudexy XR suddenly may cause serious problems. If you have epilepsy and you stop taking Qudexy XR suddenly, you may have seizures that do not stop. Your healthcare provider will tell you how to stop taking Qudexy XR slowly.
- Your healthcare provider may do blood tests while you take Qudexy XR.
What should I avoid while taking Qudexy XR?
- You should not drink alcohol while taking Qudexy XR. Qudexy XR and alcohol can affect each other causing side effects such as sleepiness and dizziness.
- Do not drive a car or operate machinery until you know how Qudexy XR affects you. Qudexy XR can slow your thinking and motor skills and may affect vision.
What are the possible side effects of Qudexy XR?
Qudexy XR may cause serious side effects, including:
See “What is the most important information I should know about Qudexy XR?”
- High blood ammonia levels. High ammonia in the blood can affect your mental activities, slow your alertness, make you feel tired, or cause vomiting. This has happened when Qudexy XR is taken with a medicine called valproic acid (Depakene and Depakote).
- Kidney stones. Drink plenty of fluids when taking Qudexy XR to decrease your chances of getting kidney stones.
- Low body temperature. Taking Qudexy XR when you are also taking valproic acid can cause a drop-in body temperature to less than 95°F, or can cause tiredness, confusion, or coma.
- Effects on thinking and alertness. Qudexy XR may affect how you think, and cause confusion, problems with concentration, attention, memory, or speech. Qudexy XR may cause depression or mood problems, tiredness, and sleepiness.
- Dizziness or loss of muscle coordination.
Call your healthcare provider right away if you have any of the symptoms above.
The most common side effects of Qudexy XR include:
- tingling of the arms and legs (paresthesia)
- not feeling hungry
- weight loss
- nervousness
- nausea
- speech problems
- tiredness
- dizziness
- sleepiness/drowsiness
- a change in the way foods taste
- upper respiratory tract infection
- decreased feeling or sensitivity, especially in the skin
- slow reactions
- difficulty with memory
- fever
- abnormal vision
- diarrhea
- pain in the abdomen
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects of Qudexy XR. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Upsher-Smith Laboratories, LLC at 1-855-899-9180.
General information about the safe and effective use of Qudexy XR
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Qudexy XR for a condition for which it was not prescribed. Do not give Qudexy XR to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Qudexy XR that is written for health professionals.
How should I store Qudexy XR?
- Store Qudexy XR capsules at room temperature between 68° to 77°F (20° to 25°C).
- Keep Qudexy XR in a tightly closed container.
- Keep Qudexy XR dry and away from moisture.
- Keep Qudexy XR and all medicines out of the reach of children.
What are the ingredients in Qudexy XR?
Active ingredient: topiramate
Inactive ingredients: microcrystalline cellulose, hypromellose 2910, ethylcellulose, diethyl phthalate, titanium dioxide, black iron oxide, red iron oxide and/or yellow iron oxide, black pharmaceutical ink, and white pharmaceutical ink (200 mg only).
Label
PRINCIPAL DISPLAY PANEL – 100 MG CAPSULE BOTTLE LABEL
- NDC 0245-1074-30
- Once-Daily Dosing
- Qudexy® XR
(topiramate) extended-release capsules - 100 mg
- PHARMACIST: Dispense the Medication
Guide provided separately to each patient. - 30 Capsules
Rx only

PRINCIPAL DISPLAY PANEL – 200 MG CAPSULE BOTTLE LABEL
- NDC 0245-1073-30
- Once-Daily Dosing
- Qudexy® XR
(topiramate) extended-release capsules - 200 mg
- PHARMACIST: Dispense the Medication
Guide provided separately to each patient. - 30 Capsules
Rx only

SRC: NLM .

