Perforomist
Generic name: formoterol (inhalation)
Drug class: Adrenergic bronchodilators
Medically reviewed by A Ras MD.
What is Perforomist?
Perforomist Inhalation Solution is a long-acting beta2 adrenergic agonist (LABA) used to control the symptoms of chronic obstructive pulmonary disease (COPD) in adults with COPD. COPD is a chronic lung disease that includes chronic bronchitis, emphysema, or both. Perforomist Inhalation Solution is only for use with a nebulizer.
LABA medicines such as Perforomist Inhalation Solution help the muscles around the airways in your lungs stay relaxed to prevent symptoms, such as wheezing, cough, chest tightness, and shortness of breath. Perforomist Inhalation Solution is for long-term use and should be taken 2 times each day, to improve the symptoms of COPD for better breathing. Perforomist Inhalation Solution is not used to treat sudden symptoms of COPD.
Perforomist is not for the treatment of asthma. It is not known if Perforomist Inhalation Solution is safe and effective in people with asthma. Perforomist Inhalation Solution should not be used in children. It is not known if Perforomist Inhalation Solution is safe and effective in children.
Description
PERFOROMIST (formoterol fumarate) Inhalation Solution is supplied as 2 mL of formoterol fumarate inhalation solution packaged in a 2.5 mL single-use low-density polyethylene vial and overwrapped in a foil pouch. Each vial contains 2 mL of a clear, colorless solution composed of formoterol fumarate dihydrate, USP equivalent to 20 mcg of formoterol fumarate in an isotonic, sterile aqueous solution containing sodium chloride, pH adjusted to 5.0 with citric acid and sodium citrate.
The active component of PERFOROMIST Inhalation Solution is formoterol fumarate dihydrate, USP, a racemate. Formoterol fumarate dihydrate is a beta2-adrenergic bronchodilator. Its chemical name is (±)-2-hydroxy-5-[(1RS)-1-hydroxy-2-[[(1RS)-2-(4-methoxyphenyl)-1-methylethyl]-amino]ethyl]formanilide fumarate dihydrate; its structural formula is:
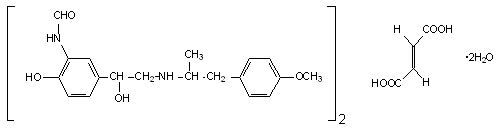
Formoterol fumarate dihydrate, USP has a molecular weight of 840.92 and its empirical formula is (C19H24N2O4)2•C4H4O4•2H2O. Formoterol fumarate dihydrate, USP is a white to yellowish crystalline powder, which is freely soluble in glacial acetic acid, soluble in methanol, sparingly soluble in ethanol and isopropanol, slightly soluble in water, and practically insoluble in acetone, ethyl acetate, and diethyl ether.
PERFOROMIST Inhalation Solution does not require dilution prior to administration by nebulization. Like all other nebulized treatments, the amount delivered to the lungs will depend on patient factors and the nebulization system used and its performance.
Using the PARI-LC Plus® nebulizer (with a facemask or mouthpiece) connected to a PRONEB® Ultra compressor under in vitro conditions, the mean delivered dose from the mouthpiece was approximately 7.3 mcg (37% of label claim). The mean nebulizer flow rate was 4 LPM and the nebulization time was 9 minutes. PERFOROMIST Inhalation Solution should be administered from a standard jet nebulizer at adequate flow rates via a facemask or mouthpiece.
Mechanism of Action
Formoterol fumarate is a long-acting, beta2-adrenergic receptor agonist (beta2-agonist). Inhaled formoterol fumarate acts locally in the lung as a bronchodilator. In vitro studies have shown that formoterol has more than 200-fold greater agonist activity at beta2-receptors than at beta1-receptors. Although beta2-receptors are the predominant adrenergic receptors in bronchial smooth muscle and beta1-receptors are the predominant receptors in the heart, there are also beta2-receptors in the human heart comprising 10% to 50% of the total beta-adrenergic receptors. The precise function of these receptors has not been established, but they raise the possibility that even highly selective beta2-agonists may have cardiac effects.
The pharmacologic effects of beta2-adrenoceptor agonist drugs, including formoterol, are at least in part attributable to stimulation of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3′, 5′-adenosine monophosphate (cyclic AMP). Increased cyclic AMP levels cause relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
In vitro tests show that formoterol is an inhibitor of the release of mast cell mediators, such as histamine and leukotrienes, from the human lung. Formoterol also inhibits histamine-induced plasma albumin extravasation in anesthetized guinea pigs and inhibits allergen-induced eosinophil influx in dogs with airway hyper-responsiveness. The relevance of these in vitro and animal findings to humans with COPD is unknown.
Who should not take Perforomist?
- Do not use Perforomist Inhalation Solution if you have asthma.
What should I tell my healthcare provider before taking Perforomist?
Before using Perforomist Inhalation Solution tell your healthcare provider about all of your medical conditions, including if you:
- have heart problems
- have high blood pressure
- have diabetes
- have seizures
- have thyroid problems
- have liver problems
- are pregnant or plan to become pregnant. It is not known if the medicine formoterol in Perforomist Inhalation Solution passes into breast milk and if it can harm your baby.
- are breastfeeding or plan to breastfeed. It is not known if the medicine formoterol in Perforomist Inhalation Solution passes into breast milk and if it can harm your baby.
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins and herbal supplements. Perforomist Inhalation Solution and certain other medicines may interact with each other. This may cause serious side effects.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine.
How should I take Perforomist?
Read the step-by-step instructions for using Perforomist Inhalation Solution that come with Perforomist.
- Use Perforomist Inhalation Solution exactly as prescribed. One unit dose vial of Perforomist Inhalation Solution is one dose. The usual dose of Perforomist Inhalation Solution is 1 unit dose vial, 2 times each day (morning and evening) breathed in through your nebulizer machine. The 2 doses should be about 12 hours apart. Do not use more than 2 vials of Perforomist Inhalation Solution a day.
- Do not mix other medicines with Perforomist Inhalation Solution in your nebulizer machine.
- If you miss a dose of Perforomist Inhalation Solution, just skip that dose. Take your next dose at your usual time. Do not take 2 doses at one time.
- While you are using Perforomist Inhalation Solution 2 times each day:
- do not use other medicines that contain a long-acting beta2-agonist (LABA) for any reason.
- do not use your short-acting beta2-agonist medicine on regular basis (four times a day).
- Perforomist Inhalation Solution does not relieve sudden symptoms of COPD. Always have a rescue inhaler medicine with you to treat sudden symptoms. If you do not have a rescue inhaler medicine, call your healthcare provider to have one prescribed for you.
- Do not stop using Perforomist Inhalation Solution or other medicines to control or treat your COPD unless told to do so by your healthcare provider because your symptoms might get worse. Your healthcare provider will change your medicines as needed.
- Do not use Perforomist Inhalation Solution:
- more often than prescribed,
- more medicine than prescribed for you, or
- with other LABA medicines
Call your healthcare provider or get emergency medical care right away if your breathing problems worsen with Perforomist Inhalation Solution, you need to use your rescue medicine more often than usual, or your rescue medicine does not work as well for you to relieve your symptoms.
What are the possible side effects of Perforomist?
Perforomist Inhalation Solution can cause serious side effects, including:
- people with asthma who take long-acting beta2 adrenergic agonist (LABA) medicines, such as Perforomist Inhalation Solution, without also using a medicine called an inhaled corticosteroid, have an increased risk of serious problems from asthma, including being hospitalized, needing a tube placed in their airway to help them breathe, or death.
- Call your healthcare provider if breathing problems worsen over time while using Perforomist Inhalation Solution. You may need a different treatment.
- Get emergency medical care if:
- breathing problems worsen quickly
- you use your rescue inhaler medicine, but it does not relieve your breathing problems
- COPD symptoms that get worse over time. If your COPD symptoms worsen over time, do not increase your dose of Perforomist Inhalation Solution, instead call your healthcare provider.
- using too much of a LABA medicine may cause:
- chest pain
- fast and irregular heartbeat
- tremor
- increased blood pressure
- headache
- nervousness
- sudden shortness of breath immediately after use of Perforomist Inhalation Solution. Sudden shortness of breath may be life-threatening. If you have sudden breathing problems immediately after inhaling your medicine, call your healthcare provider or go to nearest hospital emergency room right away.
- effects on your heart, including fast or irregular heartbeat, chest pain or increased blood pressure.
- changes in laboratory levels, including high levels of blood sugar (hyperglycemia) and low levels of potassium (hypokalemia), which may cause symptoms of muscle spasm, muscle weakness or abnormal heart rhythm.
- serious allergic reactions including rash, hives, swelling of the face, mouth, and tongue, and breathing problems. Call your healthcare provider or get emergency medical care if you get any symptoms of a serious allergic reaction.
Common side effects of Perforomist Inhalation Solution include:
- headache
- muscle cramps
- dizziness
- tremor
- nausea
- tiredness
- nervousness
- vomiting
- trouble sleeping
- dry mouth
- diarrhea
Tell your healthcare provider if you get any side effect that bothers you or that does not go away.
These are not all the possible side effects of Perforomist Inhalation Solution. For more information, ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Perforomist
Medicines are sometimes prescribed for purposes that are not listed in a Patient Information leaflet. Do not use Perforomist Inhalation Solution for a condition for which it was not prescribed. Do not give Perforomist Inhalation Solution to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your health care provider or pharmacist for information about Perforomist Inhalation Solution that is written for health professionals.
How should I store Perforomist?
- Store Perforomist Inhalation Solution in a refrigerator between 36° to 46°F (2° to 8°C) in the protective foil pouch. Protect from light and heat. Do not open a sealed pouch until you are ready to use a dose of Perforomist Inhalation Solution. After a sealed pouch is opened, Perforomist Inhalation Solution must be used right away. Perforomist Inhalation Solution may be used directly from the refrigerator.
- Perforomist Inhalation Solution may also be stored at room temperature between 68ºF to 77ºF (20º to 25ºC) for up to 3 months (90 days). If stored at room temperature, discard Perforomist Inhalation Solution if it is not used after 3 months or if past the expiration date, whichever is sooner. Space is provided on the packaging to record dispense date and use by date.
- Do not use Perforomist Inhalation Solution after the expiration date provided on the foil pouch and vial.
- Perforomist Inhalation Solution should be colorless. Discard Perforomist Inhalation Solution if it is not colorless.
Keep Perforomist Inhalation Solution and all medicines out of the reach of children.
What are the ingredients in Perforomist?
Active ingredient: formoterol
Inactive ingredients: sodium chloride, citric acid and sodium citrate
Label
PRINCIPAL DISPLAY PANEL – 20 MCG/2 ML
- NDC 49502-605-30
- Perforomist®
(formoterol fumarate) INHALATION SOLUTION
20 mcg/2 mL vial - Patient Information for Patients Enclosed
- Sterile Unit Dose Vials – Individually Wrapped – For Oral Inhalation Only
SRC: NLM .
