Paxil CR
Generic name: paroxetine
Brand names: Brisdelle, Paxil, Paxil CR, Pexeva
Drug class: Selective serotonin reuptake inhibitors
What is Paxil CR?
PAXIL CR is a prescription medicine used in adults to treat a certain type of depression called Major Depressive Disorder (MDD), Panic Disorder, Social Anxiety Disorder (SAD), Premenstrual Dysphoric Disorder (PMDD)
Description
PAXIL CR, contains paroxetine hydrochloride, an SSRI. It is the hydrochloride salt of a phenylpiperidine compound identified chemically as (-)-trans-4R-(4′-fluorophenyl)-3S-[(3′,4′-methylenedioxyphenoxy) methyl] piperidine hydrochloride hemihydrate and has the empirical formula of C19H20FNO3·HCl·1/2H2O. The molecular weight is 374.8 g/mol (329.4 g/mol as free base). The structural formula of paroxetine hydrochloride is:
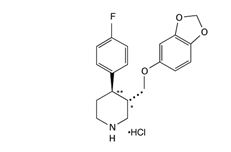
Paroxetine hydrochloride is an odorless, off‑white powder, having a melting point range of 120° C to 138°C and a solubility of 5.4 mg/mL in water.
PAXIL CR tablets are intended for oral administration. Each extended‑release tablet contains 12.5 mg, 25 mg, or 37.5 mg paroxetine equivalent to 14.25 mg, 28.51 mg, or 42.76 mg of paroxetine hydrochloride, respectively. One layer of the tablet consists of a degradable barrier layer and the other contains the active material in a hydrophilic matrix.
Inactive ingredients consist of glyceryl behenate, hypromellose, lactose monohydrate, magnesium stearate, methacrylic acid copolymer type C, polyethylene glycols, polysorbate 80, polyvinylpyrrolidone, silicon dioxide, sodium lauryl sulfate, talc, titanium dioxide, triethyl citrate and the following colorants: D&C Red No. 30 aluminum lake (25 mg), D&C Yellow No. 10 aluminum lake (12.5 mg), FD&C Blue No. 2 aluminum lake (37.5 mg), FD&C Yellow No. 6 aluminum lake (12.5 mg), red ferric oxide (25 mg) and Yellow ferric oxide (12.5 mg and 37.5 mg).
Mechanism of Action
The mechanism of action of paroxetine in the treatment of major depressive disorder (MDD), panic disorder (PD), social anxiety disorder (SAD), and premenstrual dysphoric disorder (PMDD) is unknown, but is presumed to be linked to potentiation of serotonergic activity in the central nervous system resulting from inhibition of neuronal reuptake of serotonin (5-HT).
What is the most important information I should know about Paxil CR?
Paxil CR can cause serious side effects, including:
- Increased risk of suicidal thoughts or actions. Antidepressant medicines may increase suicidal thoughts and actions in some children and young adults within the first few months of treatment or when the dose is changed. Paxil CR is not for use in people younger than 18 years of age.
How can I watch for and try to prevent suicidal thoughts and actions?
- Depression or other serious mental illnesses are the most important causes of suicidal thoughts and actions.
- Pay close attention to any changes, especially sudden changes in mood, behavior, thoughts or feelings or if you develop suicidal thoughts or actions. This is very important when an antidepressant medicine is started or when the dose is changed.
- Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts or feelings or if you develop suicidal thoughts or actions.
- Keep all follow-up visits with your healthcare provider as scheduled. Call your healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call your healthcare provider or get emergency medical help right away if you have any of the following symptoms, especially if they are new, worse, or worry you:
- attempts to commit suicide
- acting aggressive or violent
- new or worse depression
- feeling agitated, restless, angry, or irritable
- an increase in activity and talking more than what is normal for you
- acting on dangerous impulses
- thoughts about suicide or dying
- new or worse anxiety or panic attacks
- trouble sleeping
- other unusual changes in behavior or mood
Who should not take Paxil CR?
Do not take Paxil CR if you:
- take a monoamine oxidase inhibitor (MAOI)
- have stopped taking an MAOI in the last 14 days
- are being treated with the antibiotic linezolid or intravenous methylene blue
- are taking thioridazine
- are taking pimozide
- are allergic to paroxetine or any of the ingredients in Paxil CR. See the end of this Medication Guide for a complete list of ingredients in Paxil CR.
Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI or one of these medicines, including intravenous methylene blue.
Do not start taking an MAOI for at least 14 days after you stop treatment with PAXIL CR.
What should I tell my healthcare provider before taking Paxil CR?
Before taking Paxil CR, tell your healthcare provider about all your medical conditions, including if you:
- have heart problems
- have or had bleeding problems
- have, or have a family history of bipolar disorder, mania or hypomania
- have or had seizures or convulsions
- have glaucoma (high pressure in the eye)
- have low sodium levels in your blood
- have bone problems
- have kidney or liver problems
- are pregnant or plan to become pregnant. Paxil CR may harm your unborn baby. Talk to your healthcare provider about the risks to your unborn baby if you take Paxil CR during pregnancy. Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with Paxil CR.
- are breastfeeding or plan to breastfeed. Paxil CR passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with Paxil CR.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Paxil CR and some other medicines may affect each other causing possible serious side effects. Paxil CR may affect the way other medicines work and other medicines may affect the way Paxil CR works.
Especially tell your healthcare provider if you take:
- medicines used to treat migraine headaches called triptans
- tricyclic antidepressants
- fentanyl
- lithium
- tramadol
- tryptophan
- buspirone
- amphetamines
- St. John’s Wort
- medicines that can affect blood clotting such as aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), or warfarin
- diuretics
- tamoxifen
Ask your healthcare provider if you are not sure if you are taking any of these medicines. Your healthcare provider can tell you if it is safe to take Paxil CR with your other medicines.
Do not start or stop any other medicines during treatment with PAXIL CR without talking to your healthcare provider first. Stopping Paxil CR suddenly may cause you to have serious side effects. See, “What are the possible side effects of Paxil CR?”
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take Paxil CR?
- Take Paxil CR exactly as your healthcare provider tell you to. Your healthcare provider may need to change the dose of Paxil CR until it is the right dose for you.
- Take Paxil CR 1 time each day in the morning.
- Paxil CR may be taken with or without food.
- Swallow Paxil CR tablets whole. Do not chew or crush Paxil CR tablets.
- If you take too much Paxil CR, call your poison control center at 1-800-222-1222 or got to the nearest hospital emergency room right away.
What are the possible side effects of Paxil CR?
Paxil CR may cause serious side effects, including:
- See, “What is the most important information I should know about Paxil CR?”
- Serotonin syndrome. A potentially life-threatening problem called serotonin syndrome can happen when you take Paxil CR with certain other medicines. See, “Who should not take Paxil CR?” Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of the following signs and symptoms of serotonin syndrome:
- agitation
- seeing or hearing things that are not real (hallucinations)
- confusion
- coma
- fast heart beat
- changes in blood pressure
- dizziness
- sweating
- flushing
- high body temperature (hyperthermia)
- shaking (tremors), stiff muscles, or muscle twitching
- loss of coordination
- seizures
- nausea, vomiting, diarrhea
- Medicine interactions. Taking Paxil CR with certain other medicines including thioridazine and pimozide may increase the risk of developing a serious heart problem called QT prolongation.
- Abnormal bleeding. Taking Paxil CR with aspirin, NSAIDs, or blood thinners may add to this risk. Tell your healthcare provider about any unusual bleeding or bruising.
- Manic episodes. Manic episodes may happen in people with bipolar disorder who take Paxil CR. Symptoms may include:
- greatly increased energy
- racing thoughts
- unusually grand ideas
- talking more or faster than usual
- severe problems sleeping
- reckless behavior
- excessive happiness or irritability
- Discontinuation syndrome. Suddenly stopping Paxil CR may cause you to have serious side effects. Your healthcare provider may want to decrease your dose slowly. Symptoms may include:
- nausea
- sweating
- changes in mood
- irritability and agitation
- dizziness
- electric shock feeling (paresthesia)
- tremor
- anxiety
- confusion
- headache
- tiredness
- problems sleeping
- ringing in your ears (tinnitus)
- seizures
- Seizures (convulsions).
- Eye problems (angle-closure glaucoma). Paxil CR may cause a type of eye problem called angle-closure glaucoma in people with certain other eye conditions. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
- Low sodium levels in your blood (hyponatremia). Low sodium levels in your blood that may be serious and may cause death, can happen during treatment with Paxil CR. Elderly people and people who take certain medicines may be at a greater risk for developing low sodium levels in your blood. Signs and symptoms may include:
- headache
- difficulty concentrating
- memory changes
- confusion
- weakness and unsteadiness on your feet which can lead to fallsIn more severe or more sudden cases, signs and symptoms include:
- seeing or hearing things that are not real (hallucinations)
- fainting
- seizures
- coma
- stopping breathing (respiratory arrest)
- Bone fractures.
The most common side effects Paxil CR include:
- male and female sexual function problems
- blurred vision
- weakness (asthenia)
- constipation
- decreased appetite
- diarrhea
- dizziness
- dry mouth
- problems sleeping
- nausea
- sleepiness
- sweating
- tremor
These are not all the possible side effects of Paxil CR.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Paxil CR
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take Paxil CR for a condition for which it was not prescribed. Do not give Paxil CR to other people, even if they have the same symptoms that you have. It may harm them. You may ask your healthcare provider or pharmacist for information about Paxil CR that is written for healthcare professionals.
How should I store Paxil CR?
- Store Paxil CR at room temperature between 68°F to 77°F (20°C to 25°C).
Keep Paxil CR and all medicines out of the reach of children.
What are the ingredients in Paxil CR?
Active ingredient: paroxetine hydrochloride
Inactive ingredients: glyceryl behenate, hypromellose, lactose monohydrate, magnesium stearate, methacrylic acid copolymer type C, polyethylene glycols, polysorbate 80, polyvinylpyrrolidone, silicon dioxide, sodium lauryl sulfate, talc, titanium dioxide, triethyl citrate and the following colorants: D&C Red No. 30 aluminum lake (25 mg), D&C Yellow No. 10 aluminum lake (12.5 mg), FD&C Blue No. 2 aluminum lake (37.5 mg), FD&C Yellow No. 6 aluminum lake (12.5 mg), red ferric oxide (25 mg) and Yellow ferric oxide (12.5 mg and 37.5 mg).
Label
PRINCIPAL DISPLAY PANEL
- Principal Display Panel
- NDC 60505-3668-3
- PAXIL CR®
- PAROXETINE HCl
- CONTROLLED-RELEASE TABLETS
- 12.5 mg
- 30 tablets
- Rx only
- Federal Law requires dispensing of Paxil CR® with the Medication Guide provided with this bottle.
- Store at or below 25°C (77°F) [see USP].
- Each controlled-release tablet contains paroxetine hydrochloride equivalent to 12.5 mg paroxetine.
- Dosage: See accompanying prescribing information.
- Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser.
- Manufactured by:
- GlaxoSmithKline
- RTP, NC 27709
- Distributed by:
- Apotex Corp.
- Weston, FL 33326
- Made in Canada

PRINCIPAL DISPLAY PANEL
- Principal Display Panel
- NDC 60505-3669-3
- PAXIL CR®
- PAROXETINE HCl
- CONTROLLED-RELEASE TABLETS
- 25 mg
- 30 tablets
- Rx only
- Federal Law requires dispensing of Paxil CR® with the Medication Guide provided with this bottle.
- Store at or below 25°C (77°F) [see USP].
- Each controlled-release tablet contains paroxetine hydrochloride equivalent to 25 mg paroxetine.
- Dosage: See accompanying prescribing information.
- Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser.
- Manufactured by:
- GlaxoSmithKline
- RTP, NC 27709
- Distributed by:
- Apotex Corp.
- Weston, FL 33326
- Made in Canada


PRINCIPAL DISPLAY PANEL
- Principal Display Panel
- NDC 60505-3670-3
- PAXIL CR®
- PAROXETINE HCl
- CONTROLLED-RELEASE TABLETS
- 37.5 mg
- 30 tablets
- Rx only
- Federal Law requires dispensing of Paxil CR® with the Medication Guide provided with this bottle.
- Store at or below 25°C (77°F) [see USP].
- Each controlled-release tablet contains paroxetine hydrochloride equivalent to 37.5 mg paroxetine.
- Dosage: See accompanying prescribing information.
- Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser.
- Manufactured by:
- GlaxoSmithKline
- RTP, NC 27709
- Distributed by:
- Apotex Corp.
- Weston, FL 33326
- Made in Canada


SRC: NLM .
