Iclusig
Generic name: ponatinib
Drug classes: BCR-ABL tyrosine kinase inhibitors, Multikinase inhibitors, VEGF/VEGFR inhibitors
Medically reviewed by A Ras MD.
What is Iclusig?
Iclusig is a prescription medicine used to treat adults who have chronic phase chronic myeloid leukemia (CML) who did not tolerate or no longer benefit from treatment with at least 2 prior kinase inhibitor medicines, accelerated phase or blast phase CML, or Philadelphia chromosome positive acute lymphoblastic leukemia (Ph+ ALL) who cannot receive any other kinase inhibitor medicines
It is used to treat a specific type of abnormal gene (T315I-positive) chronic phase, accelerated phase, or blast phase CML, or T315I-positive Ph+ ALL
Iclusig is not for use to treat people with newly diagnosed chronic phase CML.
It is not known if Iclusig is safe and effective in children.
Description
Ponatinib is a kinase inhibitor. The chemical name for ponatinib hydrochloride is 3-(imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N-{4-[(4-methylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl}benzamide hydrochloride. The molecular formula is C29H28ClF3N6O which corresponds to a formula weight of 569.02 g/mol. Its structure is shown below:
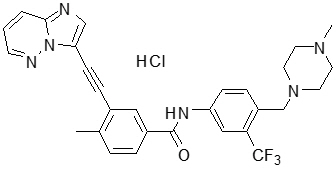
Ponatinib HCl is an off-white to yellow powder with pKa of 2.77 and 7.8. The solubility of ponatinib in pH 1.7, 2.7, and 7.5 buffers is 7790 mcg/mL, 3.44 mcg/mL, and 0.16 mcg/mL, respectively, indicating a decrease in solubility with increasing pH. Each tablet for oral administration contains 10 mg, 15 mg, 30 mg or 45 mg of ponatinib equivalent to 10.68 mg, 16.03 mg, 32.05 mg, and 48.08 mg of ponatinib hydrochloride with the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (type B), colloidal silicon dioxide, magnesium stearate and a tablet coating. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol, and titanium dioxide.
Mechanism of Action
Ponatinib is a kinase inhibitor. Ponatinib inhibited the in vitro tyrosine kinase activity of ABL and T315I mutant ABL with IC50 concentrations of 0.4 nM and 2.0 nM, respectively. Ponatinib inhibited the in vitro activity of additional kinases with IC50 concentrations between 0.1 nM and 20 nM, including members of the VEGFR, PDGFR, FGFR, EPH receptors and SRC families of kinases, and KIT, RET, TIE2, and FLT3. Ponatinib inhibited the in vitro viability of cells expressing native or mutant BCR-ABL, including T315I. In mice, treatment with ponatinib reduced the size of tumors expressing native or T315I mutant BCR-ABL when compared to controls.
What is the most important information I should know about Iclusig?
Iclusig can cause serious side effects, including:
- Blood clots or blockage in your blood vessels (arteries and veins). Blood clots or blockage in your blood vessels may lead to heart attack, stroke, or death. A blood clot or blockage in your blood vessels can prevent proper blood flow to your heart, brain, bowels (intestines), legs, eyes, and other parts of your body. You may need emergency surgery or treatment in a hospital. Get medical help right away if you get any of the following symptoms:
- chest pain or pressure
- pain in your arms, legs, back, neck or jaw
- shortness of breath
- numbness or weakness on one side of your body
- leg swelling
- trouble talking
- headache
- dizziness
- severe stomach area pain
- decreased vision or loss of vision
Blood clots or blockage in your blood vessels can happen in people with or without risk factors for heart and blood vessel disease, including people 50 years of age or younger. The most common risk factors for these problems are a history of high blood pressure (hypertension), high cholesterol, and heart disease. Blood clots or blockages in your blood vessels happen more often in people as they get older, and in people with a history of decreased blood flow, high blood pressure, diabetes, or high cholesterol.
- Heart problems. Iclusig can cause heart problems, including heart failure which can be serious and may lead to death. Heart failure means your heart does not pump blood well enough. Iclusig can also cause irregular, slow, or fast heartbeats and heart attack. Your healthcare provider will check you for heart problems during your treatment with Iclusig. Get medical help right away if you get any of the following symptoms: shortness of breath, chest pain, fast or irregular heartbeats, dizziness, or feel faint.
- Liver problems. Iclusig can cause liver problems, including liver failure, which can be severe and may lead to death. Your healthcare provider will do blood tests before and during your treatment with Iclusig to check for liver problems. Get medical help right away if you get any of these symptoms of liver problems during treatment:
- yellowing of your skin or the white part of your eyes
- dark “tea-colored” urine
- sleepiness
- loss of appetite
- bleeding or bruising
See “What are the possible side effects of Iclusig?” for information about side effects.
What should I tell my healthcare provider before taking Iclusig?
Before you take Iclusig, tell your healthcare provider about all of your medical conditions, including if you:
- have a history of blood clots in your blood vessels (arteries or veins)
- have heart problems, including heart failure, irregular heartbeats, and QT prolongation
- have diabetes
- have a history of high cholesterol
- have liver problems
- have had inflammation of your pancreas (pancreatitis)
- have high blood pressure
- have bleeding problems
- plan to have surgery or have had a recent surgery. You should stop taking Iclusig at least 1 week before planned surgery. See “What are the possible side effects of Iclusig?”.
- are lactose (milk sugar) intolerant. Iclusig tablets contain lactose.
- eat grapefruit or drink grapefruit juice. See “How should I take Iclusig?”.
- are pregnant or plan to become pregnant. Iclusig can harm your unborn baby.
- Your healthcare provider will do a pregnancy test before you start taking Iclusig.
- You should not become pregnant during treatment with Iclusig.
- For females who can become pregnant:
- Use an effective form of birth control during treatment and for 3 weeks after your last dose of Iclusig.
- Tell your healthcare provider right away if you become pregnant or think you might be pregnant during treatment with Iclusig.
- Iclusig may affect your ability to have children. Tell your healthcare provider if this is a concern for you.
- are breastfeeding or plan to breastfeed. It is not known if Iclusig passes into your breast milk. Do not breastfeed during treatment and for 6 days after your last dose of Iclusig.
Tell your healthcare provider about all the medicines you take, including prescription medicines and over-the-counter medicines, vitamins, and herbal supplements. Iclusig and other medicines may affect each other causing side effects.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take Iclusig?
- Take Iclusig exactly as your healthcare provider tells you to take it.
- Do not change your dose or stop taking Iclusig unless your healthcare provider tells you.
- Swallow Iclusig tablets whole. Do not crush, break, cut, chew or dissolve Iclusig tablets.
- Take Iclusig with or without food.
- Do not eat grapefruit or drink grapefruit juice during treatment with Iclusig.
- If you miss a dose of Iclusig, take your next dose at your regularly scheduled time the next day. Do not take 2 doses at the same time to make up for a missed dose.
- If you take too much Iclusig, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Iclusig?
Iclusig may cause serious side effects, including:
- See “What is the most important information I should know about Iclusig?”
- High blood pressure (hypertension). Iclusig can cause new or worsening high blood pressure. Your blood pressure should be checked regularly, and any high blood pressure should be treated during treatment with Iclusig. Tell your healthcare provider right away if you get confusion, headaches, dizziness, chest pain or shortness of breath.
- Inflammation of the pancreas (pancreatitis). Tell your healthcare provider right away if you get any of the following symptoms: sudden stomach-area pain or discomfort, nausea, and vomiting. Your healthcare provider should do blood tests to check for pancreatitis during treatment with Iclusig.
- Neuropathy. Iclusig may cause damage to the nerves in your arms, brain, hands, legs, or feet (neuropathy). Tell your healthcare provider right away if you get any of these symptoms during treatment with Iclusig:
- muscle weakness, tingling, burning, pain, discomfort or loss of feeling in your hands and feet
- double vision and other problems with eyesight, trouble moving the eye, drooping of part of the face, sagging or drooping eyelids, or change in taste
- Eye problems. Serious eye problems that can lead to blindness or blurred vision may happen with Iclusig. Tell your healthcare provider right away if you get any of the following symptoms: bleeding in the eye, perceived flashes of light, light sensitivity, floaters, blurred vision, dry, inflamed, swollen, or itchy eyes, or eye pain. Your healthcare provider will monitor your vision before and during your treatment with Iclusig.
- Serious bleeding. Iclusig can cause bleeding which can be serious and may lead to death. Tell your healthcare provider right away if you get any signs of bleeding during treatment with Iclusig including:
- vomiting blood or if your vomit looks like coffee-grounds
- pink or brown urine
- red or black (looks like tar) stools
- coughing up blood or blood clots
- unusual bleeding or bruising of your skin
- menstrual bleeding that is heavier than normal
- unusual vaginal bleeding
- nose bleeds that happen often
- drowsiness or difficulty being awakened
- confusion
- headache
- change in speech
- Fluid retention. Your body may hold too much fluid (fluid retention) which can be serious and may lead to death. Tell your healthcare provider right away if you get any of these symptoms during treatment with Iclusig:
- swelling of your hands, ankles, feet, face, or all over your body
- weight gain
- shortness of breath and cough
- Irregular heartbeat. Iclusig may cause an irregular heartbeat. Tell your healthcare provider right away if you experience loss of consciousness, fainting, dizziness, chest pain or palpitations.
- Low blood cell counts. Iclusig may cause low blood cell counts, which can be severe. Your healthcare provider will check your blood counts regularly during treatment with Iclusig. Tell your healthcare provider right away if you have a fever or any signs of an infection while taking Iclusig.
- Tumor Lysis Syndrome (TLS). TLS is caused by a fast breakdown of cancer cells. TLS can cause you to have:
- kidney failure and the need for dialysis treatment
- an abnormal heartbeat
Your healthcare provider may do blood tests to check for TLS. Drink plenty of water during treatment with Iclusig to help reduce your risk of getting TLS.
- Reversible Posterior Leukoencephalopathy Syndrome (RPLS – also known as Posterior Reversible Encephalopathy Syndrome). Iclusig may trigger a condition called RPLS. Call your healthcare provider right away if you get headaches, seizures, confusion, changes in vision or problems thinking.
- Wound healing problems. Wound healing problems have happened in some people who take Iclusig. Tell your healthcare provider if you plan to have any surgery before or during treatment with Iclusig.
- You should stop taking Iclusig at least 1 week before planned surgery.
- Your healthcare provider should tell you when you may start taking Iclusig again after surgery.
- A tear in your stomach or intestinal wall (perforation). Tell your healthcare provider right away if you get:
- severe pain in your stomach-area (abdomen)
- swelling of the abdomen
- high fever
The most common side effects of Iclusig include:
- skin rash
- joint pain
- stomach-area (abdomen) pain
- headache
- constipation
- dry skin
- high blood pressure
- tiredness
- swelling of your hands, ankles, feet, face, or all of your body (fluid retention and edema)
- fever
- nausea
- inflammation of the pancreas
- increase in lipase levels (a blood test done to check your pancreas)
- bleeding
- low hemoglobin in the blood (anemia)
- liver problems
- blood clots or blockage in blood vessels (arteries)
- low blood platelet counts
- low blood levels of white blood cells (including neutrophils)
Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment with Iclusig if you have certain side effects.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of Iclusig. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Iclusig
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Iclusig for a condition for which it was not prescribed. Do not give Iclusig to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Iclusig that is written for health professionals.
How should I store Iclusig?
Store Iclusig at room temperature between 68°F to 77°F (20°C to 25°C).
Keep Iclusig and all medicines out of the reach of children.
What are the ingredients in Iclusig?
Active ingredient: ponatinib
Inactive ingredients: lactose monohydrate, microcrystalline cellulose, sodium starch glycolate (type B), colloidal silicon dioxide and magnesium stearate. The tablet coating consists of talc, polyethylene glycol, polyvinyl alcohol and titanium dioxide.
Label
PRINCIPAL DISPLAY PANEL – 15 MG TABLET BOTTLE LABEL
- NDC 63020-535-30
- ICLUSIG®
(ponatinib) tablets - 15 mg
- Each tablet contains 15 mg ponatinib
equivalent to 16.03 mg ponatinib HCl - Dispense Attached Medication Guide
- 30 tablets
Rx only - Takeda

PRINCIPAL DISPLAY PANEL – 45 MG TABLET BOTTLE LABEL
- NDC 63020-534-30
- ICLUSIG®
(ponatinib) tablets - 45 mg
- Each tablet contains 45 mg ponatinib
equivalent to 48.08 mg ponatinib HCl - Dispense Attached Medication Guide
- 30 tablets
Rx only - Takeda


SRC: NLM .

