Dyanavel XR
Generic name: amphetamine
Brand names: Adzenys ER, Adzenys XR-ODT, Dyanavel XR, Evekeo, Evekeo ODT
Drug class: CNS stimulants
Medically reviewed by A Ras MD.
What is Dyanavel XR?
Dyanavel XR is a central nervous system (CNS) stimulant prescription medicine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in people 6 years of age and older. Dyanavel XR may help increase attention and decrease impulsiveness and hyperactivity in people with ADHD.
It is not known if Dyanavel XR is safe and effective in children under 6 years of age.
Dyanavel XR is a federally controlled substance (CII) because it contains amphetamine that can be a target for people who abuse prescription medicines or street drugs. Keep Dyanavel XR in a safe place to protect it from theft. Never give your Dyanavel XR to anyone else, because it may cause death or harm them. Selling or giving away Dyanavel XR may harm others and is against the law.
Description
DYANAVEL XR (amphetamine) extended-release oral suspension and DYANAVEL XR (amphetamine) extended-release tablets contain amphetamine, a CNS stimulant, in a 3.2:1 ratio of d– to l– amphetamine.
There are three active ingredients: amphetamine (complexed with sodium polystyrene sulfonate), dextroamphetamine sulfate and amphetamine aspartate. The dosage strengths are expressed in terms of amphetamine base.
DYANAVEL XR contains both immediate-release and extended-release components.
Structural Formula:
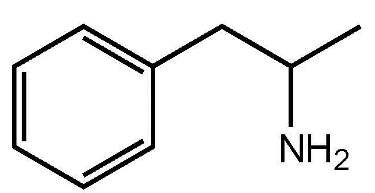
- C9H13N
- MW 135.21
- Active Ingredients:
DYANAVEL XR extended-release oral suspension 2.5 mg/mL:
- Each 1 mL contains 2 mg of amphetamine (in a 3.2 to 1 ratio of d- to l-amphetamine complexed with sodium polystyrene sulfonate), and 0.5 mg amphetamine (present as 0.5 mg of amphetamine aspartate and 0.3 mg of dextroamphetamine sulfate).
DYANAVEL XR extended-release tablets:
- Each 5 mg strength tablet contains 4 mg of amphetamine (in a 3.2 to 1 ratio of d- to l-amphetamine complexed with sodium polystyrene sulfonate), and 1 mg of amphetamine (present as 1 mg of amphetamine aspartate and 0.7 mg of dextroamphetamine sulfate).
- Each 10 mg strength tablet contains 8 mg of amphetamine (in a 3.2 to 1 ratio of d- to l-amphetamine complexed with sodium polystyrene sulfonate), and 2 mg of amphetamine (present as 2 mg amphetamine aspartate and 1.4 mg dextroamphetamine sulfate).
- Each 15 mg strength tablet contains 12 mg of amphetamine (in a 3.2 to 1 ratio of d- to l-amphetamine complexed with sodium polystyrene sulfonate), and 3 mg of amphetamine (present as 3 mg amphetamine aspartate and 2 mg dextroamphetamine sulfate).
- Each 20 mg strength tablet contains 16 mg of amphetamine (in a 3.2 to 1 ratio of d- to l-amphetamine complexed with sodium polystyrene sulfonate), and 4 mg of amphetamine (present as 4 mg amphetamine aspartate and 2.7 mg dextroamphetamine sulfate).
DYANAVEL XR extended-release oral suspension and DYANAVEL XR extended-release tablets are intended for oral administration.
Inactive Ingredients:
DYANAVEL XR extended-release oral suspension: anhydrous citric acid, bubblegum flavor, glycerin, methylparaben, modified starch, polysorbate 80, povidone, polyvinyl acetate, propylparaben, sodium lauryl sulfate, sodium polystyrene sulfonate, sucralose, triacetin and xanthan gum.
DYANAVEL XR extended-release tablets: bubblegum flavor, crospovidone, guar gum, magnesium stearate, mannitol, microcrystalline cellulose, polyvinyl acetate, povidone, silicon dioxide, sodium polystyrene sulfonate, sucralose, talc, triacetin and xanthan gum.
Mechanism of Action
Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The mode of therapeutic action in ADHD is not known.
What is the most important information I should know about Dyanavel XR?
Dyanavel XR can cause serious side effects, including:
- Abuse and dependence. Dyanavel XR, other amphetamine containing medicines, and methylphenidate have a high chance for abuse and can cause physical and psychological dependence. Your healthcare provider should check you or your child for signs of abuse and dependence before, and during, treatment with Dyanavel XR.
- Tell your healthcare provider if you or your child have ever abused or been dependent on alcohol, prescription medicines, or street drugs.
- Your healthcare provider can tell you more about the differences between physical and psychological dependence and drug addiction.
- Heart-related problems, including:
- sudden death, stroke and heart attack in adults
- sudden death in children and adolescents who have heart problems or heart defects
- increased blood pressure and heart rate
Your healthcare provider should check you or your child carefully for heart problems before starting treatment with Dyanavel XR. Tell your healthcare provider if you or your child have any heart problems, heart defects, high blood pressure, or a family history of these problems.
Your healthcare provider should check you or your child’s blood pressure and heart rate regularly during treatment with Dyanavel XR.
Call your healthcare provider or go to the nearest hospital emergency room right away if you oryour child has any signs of heart problems such as chest pain, shortness of breath, or fainting during treatment with Dyanavel XR.
- Mental (psychiatric) problems, including:
- new or worse behavior and thought problems
- new or worse bipolar illness
- new psychotic symptoms (such as hearing voices, or seeing or believing things that are not real) or new manic symptoms
Tell your healthcare provider about any mental problems you or your child have, or about a family history of suicide, bipolar illness, or depression.
Call your healthcare provider right away if you or your child have any new or worsening mental symptoms or problems during treatment with Dyanavel XR, especially hearing voices, seeing or believing things that are not real, or new manic symptoms.
Who should not take Dyanavel XR?
- allergic to amphetamine products or any of the ingredients in Dyanavel XR. See the end of this Medication Guide for a complete list of ingredients in Dyanavel XR.
- taking, or have taken within the past 14 days a medicine called a monoamine oxidase inhibitor (MAOI), including the antibiotic linezolid and the intravenous medicine methylene blue. Ask your healthcare provider or pharmacist if you are not sure if you or your child take any of these medicines.
What should I tell my healthcare provider before taking Dyanavel XR?
Before taking Dyanavel XR tell your healthcare provider about all medical conditions, including if you or your child:
- have heart problems, heart defects, or high blood pressure
- have mental problems including psychosis, mania, bipolar illness, or depression
- have circulation problems in fingers and toes
- have kidney problems
- are pregnant or plan to become pregnant. It is not known if Dyanavel XR will harm your unborn baby. Tell your healthcare provider if you become pregnant during treatment with Dyanavel XR.
- There is a pregnancy registry for females who are exposed to Dyanavel XR during pregnancy. The purpose of the registry is to collect information about the health of females exposed to Dyanavel XR and their baby. If you or your child becomes pregnant during treatment with Dyanavel XR, talk to your healthcare provider about registering with the National Pregnancy Registry or Psychostimulants at 1-866-961-2388.
- are breastfeeding or plan to breastfeed. Dyanavel XR passes into breast milk. You should not breastfeed while you are taking Dyanavel XR.
- Tell your healthcare provider about all of the medicines that you or your child takes, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Dyanavel XR and some medicines may interact with each other and cause serious side effects. Sometimes the doses of other medicines will need to be adjusted while taking Dyanavel XR.
Especially tell your healthcare provider if you or your child take
- MAOIs
- selective serotonin reuptake inhibitors (SSRIs)
- serotonin norepinephrine reuptake inhibitors (SNRIs)
- medicines used to treat migraine headaches called triptans
- tricyclic antidepressants
- tramadol
- lithium
- buspirone
- tryptophan
- St. John’s Wort
Your healthcare provider will decide whether Dyanavel XR can be taken with other medicines. Do not start any new medicine while taking Dyanavel XR without talking to your healthcare provider first.
How should I take Dyanavel XR?
- See the detailed “Instructions for Use” for information on how to give a dose of Dyanavel XR extended-release oral suspension.
- Take Dyanavel XR exactly as prescribed by your healthcare provider.
- Your healthcare provider may change the dose if needed.
- Take Dyanavel XR 1 time each day in the morning.
- Dyanavel XR can be taken with or without food.
- Dyanavel XR extended-release tablets may be chewed or swallowed whole.
- Dyanavel XR extended-release 5 mg tablets are scored and can be divided into equal parts at the score line.
- Your healthcare provider may sometimes stop Dyanavel XR treatment for a while to check ADHD symptoms.
If you or your child takes too much Dyanavel XR, call your healthcare provider or poison control center, or go to the nearest hospital emergency room right away. In case of poisoning call your poison control center at 1-800-222-1222.
What should I avoid while taking Dyanavel XR?
- You should avoid drinking alcohol during treatment with Dyanavel XR.
What are the possible side effects of Dyanavel XR?
Dyanavel XR can cause serious side effects, including:
- See “What is the most important information I should know about Dyanavel XR?”
- Slowing of growth (height and weight) in children. Children should have their height and weight checked often during treatment with Dyanavel XR . Your healthcare provider may stop the treatment with Dyanavel XR if you or your child is not growing or gaining height or weight as expected.
- Circulation problems in fingers and toes (peripheral vasculopathy, including Raynaud’s phenomenon). Signs and symptoms may include:
- fingers or toes may feel numb, cool, painful
- fingers or toes may change color from pale, to blue, to redTell your healthcare provider if you or your child have numbness, pain, skin color change, or sensitivity to temperature in your fingers or toes.Call your healthcare provider right away if you or your child have any signs of unexplained wounds appearing on fingers or toes while taking Dyanavel XR
- Serotonin syndrome. potentially life-threatening problem called serotonin syndrome may happen when Dyanavel XR is taken with certain other medicines. See, “What should I tell my healthcare provider before taking Dyanavel XR?” Stop taking Dyanavel XR and call your healthcare provider or go to the nearest hospital emergency room right away if you or your child develops any of the following signs and symptoms of serotonin syndrome:
- agitation
- fast heartbeat
- flushing
- seizures
- coma
- sweating
- loss of coordination
- confusion
- dizziness
- tremors, stiff muscles, or muscle twitching
- seeing or hearing things that are not real (hallucination)
- changes in blood pressure
- high body temperature (hyperthermia)
- nausea, vomiting, diarrhea
The most common side effects of amphetamine products include:
- dry mouth
- decreased appetite
- weight loss
- stomach pain
- nausea
- trouble sleeping
- restlessness
- extreme mood changes
- dizziness
- increased heart rate
These are not all the possible side effects of Dyanavel XR.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
General information about the safe and effective use of Dyanavel XR
Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not use Dyanavel XR for a condition for which it has not been prescribed. Do not give Dyanavel XR to other people, even if they have the same symptoms. It may harm them and it is against the law. You can ask your healthcare provider or pharmacist for information about Dyanavel XR that was written for health professionals.
How should I store Dyanavel XR?
- Store Dyanavel XR at room temperature between 68°F to 77°F (20°C to 25°C).
- Store Dyanavel XR in a safe place, like a locked cabinet.
- Dispose of remaining, unused, or expired Dyanavel XR by a medicine take-back program at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix Dyanavel XR with an undesirable, nontoxic substance such as dirt, cat litter, or used coffee grounds to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and throw away Dyanavel XR in the household trash.
Keep Dyanavel XR and all medicines out of the reach of children.
What are the ingredients in Dyanavel XR?
Active Ingredient: amphetamine
Inactive Ingredients
Extended release oral suspension: anhydrous citric acid, bubblegum flavor, glycerin, methylparaben, modified starch, polysorbate 80, povidone, polyvinyl acetate, propylparaben, sodium lauryl sulfate, sodium polystyrene sulfonate, sucralose, triacetin and xanthan gum.
Extended-release tablets: bubblegum flavor, crospovidone, guar gum, magnesium stearate, mannitol, microcrystalline cellulose, polyvinyl acetate, povidone, silicon dioxide, sodium polystyrene sulfonate, sucralose, talc, triacetin and xanthan gum.
For more information about Dyanavel XR go to http://www.dyanavelxr.com/ or call 1-732-940-0358.
Instructions for use for Dyanavel
Dyanavel XR (dī-an-uh-vel)
(amphetamine)
extended-release oral suspension, CII
Read this Instructions for Use before taking Dyanavel XR and each time you get a refill. There may be new information. This leaflet does not take the place of talking with the healthcare provider about your or your child’s medical condition or treatment.
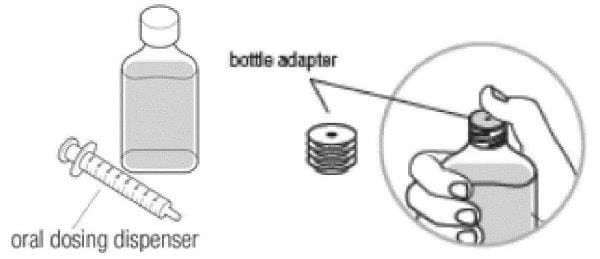
Step 1:
- Check the Dyanavel XR bottle to make sure that the bottle adapter has been inserted into the bottle by the pharmacist. Do not remove the bottle adapter.
- Check to make sure your pharmacist has given you an oral dosing dispenser.
- Tell your pharmacist if an oral dosing dispenser is not provided or the bottle adapter is missing from the neck of the bottle.
Step 2:
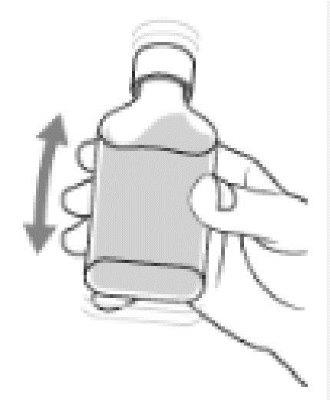
- Shake the bottle well (up and down).
Step 3:
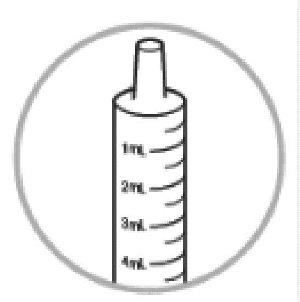
- Check the Dyanavel XR oral dosing dispenser to find the right dose in milliliters (mL) that you or your child’s healthcare provider has prescribed.
Step 4:
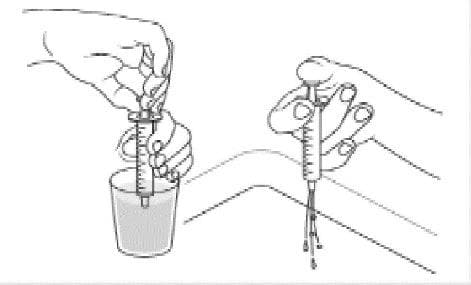
- Place the Dyanavel XR bottle upright and insert tip of the oral dosing dispenser into the bottle.
Step 5:
- Push the plunger all the way down.
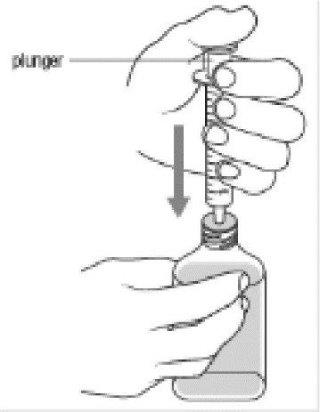
Step 6:
- With the oral dosing dispenser in place, hold the Dyanavel XR bottle with 1 hand and turn the bottle upside down. Pull the plunger down until the white end of the plunger reaches the number of mLs you need for the prescribed dose.
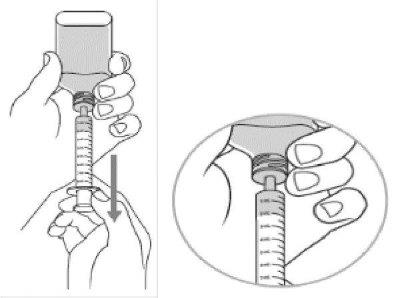
Step 7:
- Turn the bottle over and place upright on a counter top, then remove the oral dosing dispenser from the bottle adapter.
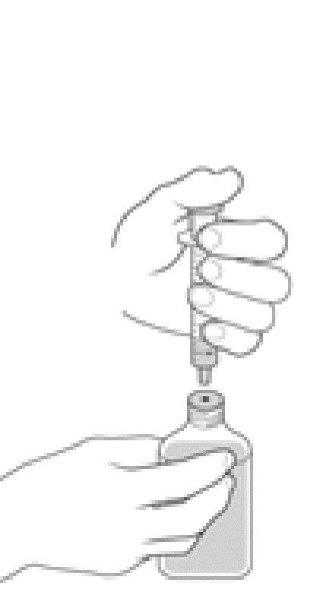
Step 8:
- Place the tip of the oral dosing dispenser into you or your child’s mouth. Point the tip toward the cheek and slowly push the plunger all the way down to give the Dyanavel XR dose.
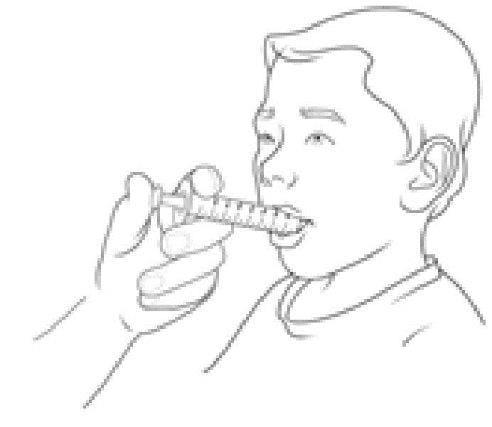
Step 9:
- Put the Dyanavel XR cap back on the bottle and close tightly.
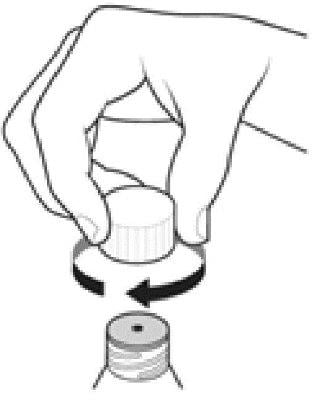
Step 10:
- Clean the oral dosing dispenser after each use by placing in the dishwasher, or by rinsing with tap water.
How should I store Dyanavel XR?
- Store Dyanavel XR at room temperature between 68°F to 77°F (20°C to 25°C).
- Store Dyanavel XR in a safe place, like a locked cabinet.
- Dispose of remaining, unused, or expired Dyanavel XR by a medicine take-back program at authorized collection sites such as retail pharmacies, hospital or clinic pharmacies, and law enforcement locations. If no take-back program or authorized collector is available, mix Dyanavel XR with an undesirable, nontoxic substance such as dirt, cat litter, or used coffee grounds to make it less appealing to children and pets. Place the mixture in a container such as a sealed plastic bag and throw away Dyanavel XR in the household trash.
Keep Dyanavel XR and all medicines out of the reach of children.
Label
PRINCIPAL DISPLAY PANEL
- NDC 24478-109-01
- Dyanavel®XR CII
- (amphetamine) extended-release Tablets
- 15 mg
- May be chewed or swallowed whole.
- 30 Tablets
- Rx only

PRINCIPAL DISPLAY PANEL
- NDC 24478-110-01
- Dyanavel® XR CII
- (amphetamine) extended-release Tablets
- 20 mg
- May be chewed or swallowed whole.
- 30 Tablets
- Rx only

SRC: NLM .
