Simponi Aria
Generic name: golimumab
Dosage form: intravenous (infusion) injection
Drug class: TNF alfa inhibitors
Medically reviewed by A Ras MD.
What is Simponi Aria?
Simponi Aria is a prescription medicine called a TNF-blocker. Simponi Aria is used to treat adults with the medicine methotrexate (MTX) to treat moderately to severely active rheumatoid arthritis (RA), Active psoriatic arthritis (PsA) in people 2 years of age and older, adults with active ankylosing spondylitis (AS), Active polyarticular juvenile idiopathic arthritis (pJIA) in people 2 years of age and older.
It is not known if Simponi Aria is safe and effective in children with PsA and pJIA under 2 years of age or in children with conditions other than PsA and pJIA.
Description
Golimumab is a human IgG1қ monoclonal antibody specific for human tumor necrosis factor alpha (TNFα) that exhibits multiple glycoforms with molecular masses of approximately 150 to 151 kilodaltons. Golimumab was created using genetically engineered mice immunized with human TNF, resulting in an antibody with human-derived antibody variable and constant regions. Golimumab is produced by a recombinant cell line cultured by continuous perfusion and is purified by a series of steps that includes measures to inactivate and remove viruses.
The SIMPONI ARIA® (golimumab) Injection is a sterile solution of the golimumab antibody supplied in a 4-mL glass vial for intravenous infusion.
SIMPONI ARIA is a preservative-free, colorless to light yellow solution with a pH of approximately 5.5. SIMPONI ARIA is not made with natural rubber latex. Each 4-mL vial of SIMPONI ARIA contains 50 mg golimumab, L-histidine (1.14 mg), L-histidine monohydrochloride monohydrate (6.42 mg), polysorbate 80 (0.6 mg), sorbitol (180 mg), and water for injection.
Mechanism of Action
Golimumab is a human monoclonal antibody that binds to both the soluble and transmembrane bioactive forms of human TNFα. This interaction prevents the binding of TNFα to its receptors, thereby inhibiting the biological activity of TNFα (a cytokine protein). There was no evidence of the golimumab antibody binding to other TNF superfamily ligands; in particular, the golimumab antibody did not bind or neutralize human lymphotoxin. Golimumab did not lyse human monocytes expressing transmembrane TNF in the presence of complement or effector cells.
Elevated TNFα levels in the blood, synovium, and joints have been implicated in the pathophysiology of several chronic inflammatory diseases such as rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. TNFα is an important mediator of the articular inflammation that is characteristic of these diseases. Golimumab modulated the in vitro biological effects mediated by TNF in several bioassays, including the expression of adhesion proteins responsible for leukocyte infiltration (E-selectin, ICAM-1 and VCAM-1) and the secretion of proinflammatory cytokines (IL-6, IL-8, G-CSF and GM-CSF). The clinical relevance of these findings is unknown.
What is the most important information I should know about Simponi Aria?
Simponi Aria is a medicine that affects your immune system. Simponi Aria can lower the ability of your immune system to fight infections. Some people have serious infections while receiving Simponi Aria, including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that spread throughout their body. Some people have died from these serious infections.
- Your doctor should test you for TB and hepatitis B before starting Simponi Aria.
- Your doctor should monitor you closely for signs and symptoms of TB during treatment with Simponi Aria.
You should not start receiving Simponi Aria if you have any kind of infection unless your doctor tells you to.
Before receiving Simponi Aria, tell your doctor if you:
- think you have an infection or have symptoms of an infection such as:
- fever, sweat, or chills
- muscle aches
- cough
- shortness of breath
- blood in phlegm
- weight loss
- warm, red, or painful skin or sores on your body
- diarrhea or stomach pain
- burning when you urinate or urinate more often than normal
- feel very tired
- are being treated for an infection.
- get a lot of infections or have infections that keep coming back.
- have diabetes, HIV, or a weak immune system. People with these conditions have a higher chance for infections.
- have TB, or have been in close contact with someone with TB.
- live, have lived, or traveled to certain parts of the country (such as the Ohio and Mississippi River valleys and the Southwest) where there is an increased chance for getting certain kinds of fungal infections (histoplasmosis, coccidioidomycosis, blastomycosis). These infections may happen or become more severe if you use Simponi Aria. Ask your doctor if you do not know if you have lived in an area where these infections are common.
- have or have had hepatitis B.
- use the medicine Orencia (abatacept), Kineret (anakinra), Actemra (tocilizumab) or Rituxan (rituximab).
After receiving Simponi Aria, call your doctor right away if you have any symptoms of an infection. Simponi Aria can make you more likely to get infections or make worse any infection that you have.
Cancer
- For children and adults receiving Tumor Necrosis Factor (TNF)-blocker medicines, including Simponi Aria, the chances of getting cancer may increase.
- There have been cases of unusual cancers in children and teenage patients receiving TNF-blocking agents.
- People with inflammatory diseases, including rheumatoid arthritis (RA), especially those with very active disease, may be more likely to get lymphoma.
- Some people receiving TNF-blockers, like Simponi Aria, developed a rare type of cancer called hepatosplenic T-cell lymphoma. This type of cancer often results in death. Most of these people were male teenagers or young men. Also, most people were being treated for Crohn’s disease or ulcerative colitis with a TNF blocker and another medicine called azathioprine or 6-mercaptopurine, (6-MP).
- Some people treated with Simponi Aria developed skin cancer. If any changes in the appearance of your skin or growths on your skin occur during or after your treatment with Simponi Aria, tell your doctor.
- You should see your doctor periodically for skin examinations, especially if you have a history of skin cancer.
What should I tell my healthcare provider before using Simponi Aria?
See “What is the most important information I should know about Simponi Aria?”.
Before starting Simponi Aria, tell your doctor about all your medical conditions, including if you:
- have an infection.
- have or have had lymphoma or any other type of cancer.
- have or have had heart failure.
- have or have had a condition that affects your nervous system, such as multiple sclerosis or Guillain-Barré syndrome.
- have a skin problem called psoriasis.
- have recently received or are scheduled to receive a vaccine. People receiving Simponi Aria should not receive live vaccines or treatment with a weakened bacteria (such as BCG for bladder cancer). People receiving Simponi Aria can receive non-live vaccines.
- have a baby and you were receiving Simponi Aria during your pregnancy. Tell your baby’s doctor before your baby receives any vaccine. Your baby may have an increased chance of getting an infection for up to 6 months after birth.
- are pregnant or plan to become pregnant. It is not known if Simponi Aria will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Simponi Aria passes into your breast milk. You and your doctor should decide if you will receive Simponi Aria or breastfeed.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially, tell your doctor if you:
- use Orencia (abatacept) or Kineret (anakinra). You should not receive Simponi Aria while you are also receiving Orencia (abatacept) or Kineret (anakinra).
- use other TNF-blocker medicines, including Remicade (infliximab), Humira (adalimumab), Enbrel (etanercept), or Cimzia (certolizumab pegol).
- receive Rituxan (rituximab) or Actemra (tocilizumab).
Ask your doctor or pharmacist for a list of these medicines if you are not sure.
Keep a list of all your medicines with you to show your doctor and pharmacist each time you get a new medicine.
How should I use Simponi Aria?
- Simponi Aria is prepared and given by a healthcare provider through a needle placed in your vein (infusion). The infusion is usually given in your arm and should take 30 minutes.
- Your doctor will decide how much Simponi Aria you will receive based on your weight. Your usual schedule for receiving Simponi Aria after your first treatment should be:
- 4 weeks after your first treatment
- every 8 weeks after that
- If you miss an appointment to receive Simponi Aria, make another appointment as soon as possible.
What are the possible side effects of Simponi Aria?
Simponi Aria can cause serious side effects, including:
See “What is the most important information I should know about Simponi Aria?”
Serious Infections.
- Some patients have an increased chance of getting serious infections while receiving Simponi Aria. These serious infections include TB and infections caused by viruses, fungi, or bacteria that have spread throughout the body. Some patients die from these infections. If you get an infection while receiving treatment with Simponi Aria your doctor will treat your infection and may need to stop your Simponi Aria treatment. Tell your doctor right away if you have any of the following signs of an infection while receiving or after receiving Simponi Aria:
- a fever
- feel very tired
- have a cough
- have flu-like symptoms
- warm, red, or painful skin
- Your doctor will examine you for TB and perform a test to see if you have TB. If your doctor feels that you are at risk for TB, you may be treated with medicine for TB before you begin treatment with Simponi Aria and during treatment with Simponi Aria. Even if your TB test is negative your doctor should carefully monitor you for TB infections while you are receiving Simponi Aria. People who had a negative TB skin test before receiving Simponi Aria have developed active TB. Tell your doctor if you have any of the following symptoms while receiving or after receiving Simponi Aria:
- cough that does not go away
- low grade fever
- weight loss
- loss of body fat and muscle (wasting)
Hepatitis B infection in people who carry the virus in their blood. If you are a carrier of the hepatitis B virus (a virus that affects the liver), the virus can become active while you use Simponi Aria. Your doctor should do blood tests before you start treatment with Simponi Aria and while you are receiving Simponi Aria.
- Tell your doctor if you have any of the following symptoms of a possible hepatitis B infection:
- feel very tired
- dark urine
- skin or eyes look yellow
- little or no appetite
- vomiting
- muscle aches
- clay-colored bowel movements
- fevers
- chills
- stomach discomfort
- skin rash
Heart failure, including new heart failure or worsening of heart failure that you already have can happen in people who use TNF-blocker medicines, including Simponi Aria. If you develop new or worsening heart failure with Simponi Aria, you may need to be treated in a hospital, and it may result in death.
- If you have heart failure before starting Simponi Aria, your condition should be watched closely during treatment with Simponi Aria.
- Call your doctor right away if you get new or worsening symptoms of heart failure during treatment with Simponi Aria (such as shortness of breath or swelling of your lower legs or feet, or sudden weight gain).
Nervous System Problems. Rarely, people receiving TNF-blocker medicines, including Simponi Aria, have nervous system problems such as multiple sclerosis or Guillain-Barré syndrome. Tell your doctor right away if you get any of these symptoms:
- vision changes
- weakness in your arms or legs
- numbness or tingling in any part of your body
Immune System Problems. Rarely, people receiving TNF-blocker medicines have developed symptoms that are like the symptoms of lupus. Tell your doctor if you have any of these symptoms:
- a rash on your cheeks or other parts of the body
- sensitivity to the sun
- new joint or muscle pains
- becoming very tired
- chest pain or shortness of breath
- swelling of the feet, ankles, or legs
Liver Problems. Liver problems can happen in people who receive TNF-blocker medicines, including Simponi Aria. These problems can lead to liver failure and death. Call your doctor right away if you have any of these symptoms:
- feel very tired
- skin or eyes look yellow
- poor appetite or vomiting
- pain on the right side of your stomach (abdomen)
Blood Problems. Low blood counts have been seen with Simponi Aria. Your body may not make enough blood cells that help fight infections or help stop bleeding. Symptoms include fever, bruising or bleeding easily, or looking pale. Your doctor will check your blood counts before and during treatment with Simponi Aria.
Allergic Reactions. Allergic reactions can happen in people who receive TNF-blocker medicines, including Simponi Aria. Some reactions may be serious and can be life-threatening. Some of these reactions can happen after receiving your first dose of Simponi Aria. Call your doctor right away if you have any of these symptoms of an allergic reaction:
- hives
- swollen face
- breathing trouble
- chest pain
The most common side effects of Simponi Aria include:
- upper respiratory infection (runny nose, sore throat, and hoarseness or laryngitis)
- abnormal liver tests
- decreased blood cells that fight infection
- viral infections, such as flu and cold sores in the mouth
- bronchitis
- high blood pressure
- rash
These are not all of the possible side effects of Simponi Aria.
Tell your doctor about any side effect that bothers you or does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Simponi Aria
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
You can ask your doctor or pharmacist for information about Simponi Aria that is written for health professionals.
What are the ingredients in Simponi Aria?
Active ingredient: golimumab.
Inactive ingredients: L-histidine, L-histidine monohydrochloride monohydrate, polysorbate 80, sorbitol, and water for injection. Simponi Aria i
Label
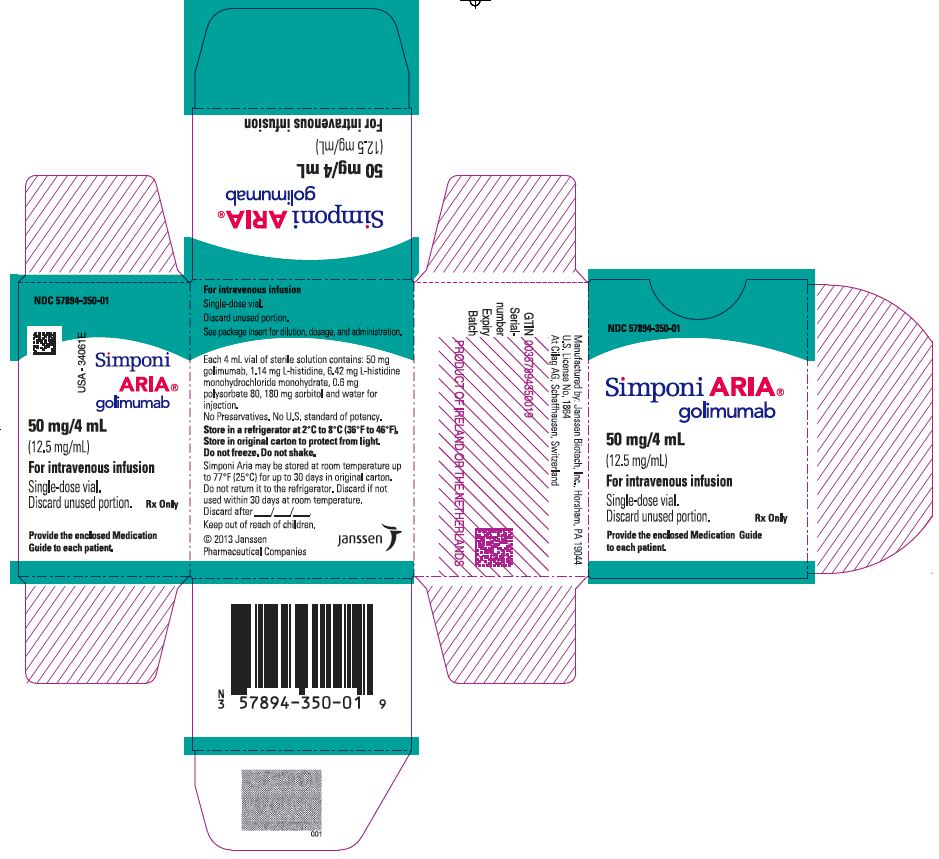
PRINCIPAL DISPLAY PANEL – 4 ML VIAL CARTON
- NDC 57894-350-01
- Simponi ARIA®
golimumab - 50 mg/4 mL
(12.5 mg/mL) - For intravenous infusion
- Single-dose vial.
Discard unused portion.
Rx Only - Provide the enclosed Medication
Guide to each patient.
 SRC: NLM .
SRC: NLM .