Samsca
Generic name: tolvaptan
Drug class: Vasopressin antagonists
Medically reviewed by A Ras MD.
What is Samsca?
Samsca is a prescription medicine used to help increase low sodium levels in the blood, in adults with conditions such as heart failure, and certain hormone imbalances. Samsca helps raise salt levels in your blood by removing extra body water as urine.
It is not known if Samsca is safe or works in children.
Description
SAMSCA contains tolvaptan, a selective vasopressin V2-receptor antagonist in tablets for oral use available in 15 mg or 30 mg strengths. Tolvaptan is (±)-4′-[(7-chloro-2,3,4,5-tetrahydro-5-hydroxy-1H-1-benzazepin-1-yl) carbonyl]-o-tolu-m-toluidide. The empirical formula is C26H25ClN2O3. Molecular weight is 448.94. The chemical structure is:
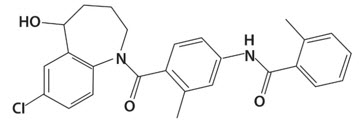
Inactive ingredients include corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate, microcrystalline cellulose, and FD&C Blue No. 2 Aluminum Lake as colorant.
Mechanism of Action
Tolvaptan is a selective vasopressin V2-receptor antagonist with an affinity for the V2-receptor that is 1.8 times that of native arginine vasopressin (AVP). Tolvaptan affinity for the V2-receptor is 29 times greater than for the V1a-receptor. When taken orally, 15 to 60 mg doses of tolvaptan antagonize the effect of vasopressin and cause an increase in urine water excretion that results in an increase in free water clearance (aquaresis), a decrease in urine osmolality, and a resulting increase in serum sodium concentrations. Urinary excretion of sodium and potassium and plasma potassium concentrations are not significantly changed. Tolvaptan metabolites have no or weak antagonist activity for human V2-receptors compared with tolvaptan.
What is the most important information I should know about Samsca?
1) Samsca may make the salt (sodium) level in your blood rise too fast. This can increase your risk of a serious condition called osmotic demyelination syndrome (ODS). ODS can lead to coma or death. ODS can also cause new symptoms such as:
- trouble speaking
- swallowing trouble or feeling like food or liquid gets stuck while swallowing
- drowsiness
- confusion
- mood changes
- trouble controlling body movement (involuntary movement) and weakness in muscles of the arms and legs
- seizures
You or a family member should tell your healthcare provider right away if you have any of these symptoms even if they begin later in treatment. Also tell your healthcare provider about any other new symptoms while taking Samsca.
You may be more at risk for ODS if you have:
- liver disease
- not eaten enough for a long period of time (malnourished)
- very low sodium level in your blood
- been drinking large amounts of alcohol for a long period of time (chronic alcoholism)
To lessen your risk of ODS while taking Samsca:
- Treatment with Samsca should be started and re-started only in a hospital, where the sodium levels in your blood can be checked closely.
- Do not take Samsca if you cannot tell if you are thirsty.
- To prevent losing too much body water (dehydration), have water available to drink at all times while taking Samsca. Unless your healthcare provider tells you otherwise, drink when you are thirsty.
- If your healthcare provider tells you to keep taking Samsca after you leave a hospital, it is important that you do not stop and re-start Samsca on your own. You may need to go back to a hospital to re-start Samsca. Talk to your healthcare provider right away if you stop taking Samsca for any reason.
- It is important to stay under the care of your healthcare provider while taking Samsca and follow their instructions.
2) Samsca may cause liver problems, including life-threatening liver failure. Samsca should not be taken for more than 30 days. Tell your doctor right away if you develop or have worsening of any of these signs and symptoms of liver problems:
- Loss of appetite, nausea, vomiting
- Fever, feeling unwell, unusual tiredness
- Itching
- Yellowing of the skin or the whites of the eyes (jaundice)
- Unusual darkening of the urine
- Right upper stomach area pain or discomfort
3) If you have autosomal dominant polycystic kidney disease (ADPKD), do not use Samsca because you should receive the medicine (tolvaptan) through a program that ensures laboratory monitoring of your liver.
Who should not take Samsca?
Do not take Samsca if:
- you are allergic to tolvaptan or any of the ingredients in Samsca. See the end of this Medication Guide for a complete list of ingredients in Samsca.
- the sodium level in your blood must be increased right away.
- you cannot replace fluids by drinking or you cannot feel if you are thirsty.
- you are dizzy, faint, or your kidneys are not working normally because you have lost too much body fluid.
- you take certain medicines. These medicines could cause you to have too much Samsca in your blood:
- the antibiotic medicines, clarithromycin (Biaxin, Biaxin XL) or telithromycin (Ketek)
- the antifungal medicines, ketoconazole (Nizoral) or itraconazole (Sporanox)
- the anti-HIV medicines, ritonavir (Kaletra, Norvir), indinavir (Crixivan), nelfinavir (Viracept), and saquinavir (Invirase)
- the antidepressant medicine, nefazodone hydrochloride
- your body is not able to make urine. Samsca will not help your condition.
What should I tell my healthcare provider before taking Samsca?
Tell your healthcare provider about all your medical conditions, including if you:
- have kidney problems and your body cannot make urine.
- have liver problems
- cannot feel if you are thirsty. See “What is the most important information I should know about Samsca?”
- have any allergies. See the end of this Medication Guide for a list of the ingredients in Samsca.
- are pregnant or plan to become pregnant. It is not known if Samsca will harm your unborn baby.
- are breast-feeding. It is not known if Samsca passes into your breast milk. You and your healthcare provider should decide if you will take Samsca or breast-feed. You should not do both.
- are taking desmopressin (dDAVP).
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Using Samsca with certain medicines could cause you to have too much Samsca in your blood. See “Who should not take Samsca?”
Samsca may affect the way other medicines work, and other medicines may affect how Samsca works.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take Samsca?
- See “What is the most important information I should know about Samsca?”
- Take Samsca exactly as prescribed by your healthcare provider.
- Take Samsca one time each day.
- You can take Samsca with or without food.
- Do not drink grapefruit juice during treatment with Samsca. This could cause you to have too much Samsca in your blood.
- Certain medicines or illnesses may keep you from drinking fluids or may cause you to lose too much body fluid, such as vomiting or diarrhea. If you have these problems, call your healthcare provider right away.
- Do not miss or skip doses of Samsca. If you miss a dose, take it as soon as you remember. If it is near the time of the next dose, skip the missed dose. Just take the next dose at your regular time. Do not take 2 doses at the same time.
- If you take too much Samsca, call your healthcare provider right away. If you take an overdose of Samsca, you may need to go to a hospital.
- If your healthcare provider tells you to stop taking Samsca, follow their instructions about limiting the amount of fluid you should drink.
What are the possible side effects of Samsca?
Samsca can cause serious side effects including:
- See “What is the most important information I should know about Samsca?”
- Loss of too much body fluid (dehydration). Tell your healthcare provider if you:
- have vomiting or diarrhea, and cannot drink normally.
- feel dizzy or faint. These may be symptoms that you have lost too much body fluid.
Call your healthcare provider right away, if you have any of these symptoms.
The most common side effects of Samsca are:
- thirst
- dry mouth
- weakness
- constipation
- making large amounts of urine and urinating often
- increased blood sugar levels
These are not all the possible side effects of Samsca. Talk to your healthcare provider about any side effect that bothers you or that does not go away while taking Samsca.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Samsca
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Samsca for a condition for which it was not prescribed. Do not give Samsca to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about Samsca. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Samsca that is written for healthcare professionals. For more information about Samsca, call 1-877-726-7220 or go to www.samsca.com.
How should I store Samsca?
Store Samsca between 59°F to 86°F (15°C to 30°C).
Keep Samsca and all medicines out of the reach of children.
What are the ingredients in Samsca?
Active ingredient: tolvaptan.
Inactive ingredients: corn starch, hydroxypropyl cellulose, lactose monohydrate, low-substituted hydroxypropyl cellulose, magnesium stearate and microcrystalline cellulose, and FD&C Blue No. 2 Aluminum Lake as colorant
Label
PRINCIPAL DISPLAY PANEL – 15 MG TABLET BLISTER PACK CARTON
- 10 Tablets
NDC 59148-020-50 - Carton contains 1 strip with 10 tablets.
- Samsca®
(tolvaptan)
tablets - 15 mg
- MUST INITIATE IN HOSPITAL
- DISPENSE THE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT - Rx only


PRINCIPAL DISPLAY PANEL – 30 MG TABLET BLISTER PACK CARTON
- 10 Tablets
NDC 59148-021-50 - Carton contains 1 strip with 10 tablets.
- Samsca®
(tolvaptan)
tablets - 30 mg
- MUST INITIATE IN HOSPITAL
- DISPENSE THE ENCLOSED
MEDICATION GUIDE TO EACH PATIENT - Rx only

SRC: NLM .
