Rebetol
Generic name: ribavirin (oral)
Drug classes: Inhaled anti-infectives, Purine nucleosides
What is Rebetol?
Rebetol is a prescription medicine used with either interferon alfa-2b or peginterferon alfa-2b to treat chronic (lasting a long time) hepatitis C infection in people 3 years and older with liver disease.
It is not known if Rebetol use for longer than 1 year is safe and will work.
It is not known if Rebetol use in children younger than 3 years old is safe and will work.
Description
REBETOL (ribavirin), is a synthetic nucleoside analogue (purine analogue). The chemical name of ribavirin is 1-β-D-ribofuranosyl-1H-1,2,4-triazole-3-carboxamide and has the following structural formula.
Ribavirin is a white, crystalline powder. It is freely soluble in water and slightly soluble in anhydrous alcohol. The empirical formula is C8H12N4O5 and the molecular weight is 244.21.
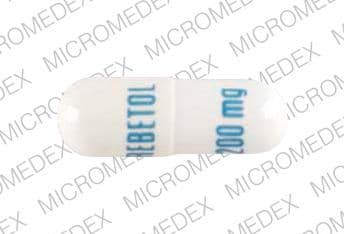
REBETOL capsules consist of a white powder in a white, opaque, gelatin capsule. Each capsule contains 200 mg ribavirin and the inactive ingredients microcrystalline cellulose, lactose monohydrate, croscarmellose sodium, and magnesium stearate. The capsule shell consists of gelatin, sodium lauryl sulfate, silicon dioxide, and titanium dioxide. The capsule is printed with edible blue pharmaceutical ink which is made of shellac, anhydrous ethyl alcohol, isopropyl alcohol, n-butyl alcohol, propylene glycol, ammonium hydroxide, and FD&C Blue #2 aluminum lake.
REBETOL oral solution is a clear, colorless to pale or light-yellow bubble gum-flavored liquid. Each milliliter of the solution contains 40 mg of ribavirin and the inactive ingredients sucrose, glycerin, sorbitol, propylene glycol, sodium citrate, citric acid, sodium benzoate, natural and artificial flavor for bubble gum #15864, and water.
What is the most important information I should know about Rebetol?
1. Rebetol may cause birth defects, miscarriage or death of your unborn baby. Do not take Rebetol if you or your sexual partner is pregnant or plan to become pregnant. Do not become pregnant during treatment or within 6 months after stopping treatment with Rebetol. You must use effective birth control during treatment with Rebetol and for 6 months after stopping treatment.
- Females must have a pregnancy test before starting Rebetol, during treatment with Rebetol, and for 6 months after the last dose of Rebetol.
- If you or your female sexual partner becomes pregnant during treatment with Rebetol or within 6 months after you stop taking Rebetol, tell your healthcare provider right away.
2. Rebetol may cause a significant drop in your red blood cell count and cause anemia in some cases. Anemia has been associated with worsening of heart problems, and in rare cases can cause a heart attack and death. Tell your healthcare provider if you have ever had any heart problems. Rebetol may not be right for you. Get medical help right away if you experience chest pain.
3. Do not take Rebetol alone to treat chronic hepatitis C infection. Rebetol should be used in combination with either interferon alfa-2b or peginterferon alfa-2b to treat chronic hepatitis C infection.
Who should not take Rebetol?
See “What is the most important information I should know about Rebetol?”
Do not take Rebetol if you have:
- ever had serious allergic reactions to the ingredients in Rebetol. See the end of this Medication Guide for a complete list of ingredients.
- certain types of hepatitis (autoimmune hepatitis).
- certain blood disorders (hemoglobinopathies).
- severe kidney disease.
- taken or currently take didanosine.
Talk to your healthcare provider before taking Rebetol if you have any of these conditions.
What should I tell my healthcare provider before taking Rebetol?
Before you take Rebetol, tell your healthcare provider if you have or ever had:
- treatment for hepatitis C that did not work for you
- breathing problems. Rebetol may cause or worsen breathing problems you already have.
- vision problems. Rebetol may cause eye problems or worsen eye problems you already have. You should have an eye exam before you start treatment with Rebetol.
- certain blood disorders such as anemia (low red blood cell count)
- high blood pressure, heart problems, or have had a heart attack. Your healthcare provider should check your blood and heart before you start treatment with Rebetol.
- thyroid problems
- liver problems other than hepatitis C infection
- human immunodeficiency virus (HIV) or any immunity problems
- mental health problems, including depression and thoughts of hurting yourself or others
- kidney problems
- an organ transplant
- diabetes. Rebetol may make your diabetes worse or harder to treat.
- any other medical condition
- are breastfeeding. It is not known if Rebetol passes into your breast milk. You and your healthcare provider should decide if you will take Rebetol or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription medicines, vitamins, and herbal supplements. Rebetol may affect the way other medicines work.
Especially tell your healthcare provider if you take didanosine or a medicine that contains azathioprine.
Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine.
How should I take Rebetol?
- Take Rebetol exactly as your healthcare provider tells you. Your healthcare provider will tell you how much Rebetol to take and when to take it.
- Take Rebetol with food.
- Take Rebetol Capsules whole. Do not open, break, or crush Rebetol Capsules before swallowing. If you cannot swallow Rebetol Capsules whole, tell your healthcare provider.
- If you miss a dose of Rebetol, take the missed dose as soon as possible during the same day. Do not double the next dose. If you have questions about what to do, call your healthcare provider.
- If you take too much Rebetol, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Rebetol?
Rebetol may cause serious side effects, including:
See “What is the most important information I should know about Rebetol?”
- Swelling and irritation of your pancreas (pancreatitis). Symptoms may include: stomach pain, nausea, vomiting, or diarrhea.
- Serious breathing problems. Difficulty breathing may be a sign of a serious lung infection (pneumonia) that can lead to death.
- Serious eye problems that may lead to vision loss or blindness.
- Dental problems. Brush your teeth well 2 times each day. Get regular dental exams. If you vomit at any time during treatment with Rebetol, rinse out your mouth well.
- Severe blood disorders. You may have an increased risk of developing severe blood disorders when Rebetol is used in combination with pegylated alpha interferons and azathioprine. Your healthcare provider should do blood tests during your treatment with Rebetol in combination with pegylated alpha interferon and azathioprine to check you for these problems.
- Growth problems in children. Weight loss and slowed growth are common in children during combination treatment with Rebetol and peginterferon alfa-2b or interferon alfa-2b. Most children will go through a growth spurt and gain weight after treatment stops. Some children may not reach the height that they were expected to have before treatment. Talk to your healthcare provider if you are concerned about your child’s growth during treatment with Rebetol and peginterferon alfa-2b or with Rebetol and interferon alfa-2b.
- Severe depression.
- Thoughts of hurting yourself or others, and suicide attempts. Adults and children who take Rebetol, especially teenagers, are more likely to have suicidal thoughts or attempt to hurt themselves while taking Rebetol. Call your healthcare provider right away or go to the nearest hospital emergency room if you have new or worse depression or thoughts about hurting yourself or others or dying.
The most common side effects of Rebetol in adults include:
- flu-like symptoms – feeling tired or weak, headache, shaking chills along with high temperature (fever), nausea, and muscle aches
- mood changes, feeling irritable
The most common side effects of Rebetol in children include:
- fever
- headache
- a decrease in blood cells that fight infection (neutropenia)
- tiredness
- decreased appetite
- vomiting
These are not all the possible side effects of Rebetol. For more information ask your healthcare provider or pharmacist.
Call your healthcare provider if you have any side effect that bothers you or that does not go away, and for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Rebetol
It is not known if treatment with Rebetol will cure hepatitis C virus infections or prevent cirrhosis, liver failure, or liver cancer that can be caused by hepatitis C virus infections. It is not known if taking Rebetol will prevent you from infecting another person with the hepatitis C virus.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Rebetol for a condition for which it was not prescribed. Do not give Rebetol to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about Rebetol that is written for health professionals.
How should I store Rebetol?
- Store Rebetol Capsules at room temperature between 68°F to 77°F (20°C to 25°C).
- Store Rebetol Oral Solution at room temperature between 68°F to 77°F (20°C to 25°C) or in the refrigerator between 36°F to 46°F (2°C to 8°C).
Keep Rebetol and all medicines out of the reach of children.
What are the ingredients in Rebetol?
Active ingredients: ribavirin
Inactive ingredients:
Capsules: microcrystalline cellulose, lactose monohydrate, croscarmellose sodium, and magnesium stearate. The capsule shell consists of gelatin, sodium lauryl sulfate, silicon dioxide, and titanium dioxide. The capsule is printed with edible blue pharmaceutical ink which is made of shellac, anhydrous ethyl alcohol, isopropyl alcohol, n-butyl alcohol, propylene glycol, ammonium hydroxide, and FD&C Blue #2 aluminum lake.
Oral solution: sucrose, glycerin, sorbitol, propylene glycol, sodium citrate, citric acid, sodium benzoate, natural and artificial flavor for bubble gum # 15864, and water.
Label
PRINCIPAL DISPLAY PANEL – 100 ML BOTTLE CARTON
- NDC 0085-1318-01
- 100 mL
- REBETOL®
(Ribavirin USP)
Oral Solution - 40
mg
per mL - ATTENTION PHARMACIST:
Must be dispensed in original
packaging with the enclosed
Medication Guide. - Rx only
- BUBBLE GUM
FLAVOR

SRC: NLM .

