Rasuvo
Generic name: methotrexate (injection)
Brand names: EmGyn Kit, Methotrexate Sodium, Preservative Free, Otrexup, Rasuvo
Drug classes: Antimetabolites, Antipsoriatics, Antirheumatics, Other immunosuppressants
Medically reviewed by A Ras MD.
What is Rasuvo?
Rasuvo is a single-dose manually-triggered auto-injector containing a prescription medicine, methotrexate. Methotrexate is used to treat certain adults with severe, active rheumatoid arthritis (RA), and children with active polyarticular juvenile idiopathic arthritis (pJIA), after treatment with other medicines including non-steroidal anti-inflammatory (NSAIDs) have been used and did not work well control the symptoms of severe, resistant, disabling psoriasis in adults when other types of treatment have been used and did not work well.
Rasuvo is available in doses of 7.5, 10, 12.5, 15, 17.5, 20, 22.5, 25, 27.5 and 30 mg. Your doctor will prescribe a different way to take methotrexate if you need to take methotrexate by mouth or in some other way. Your doctor may also change your prescription if your dose does not match the available Rasuvo doses, such as doses of less than 7.5 mg, more than 30 mg, or doses in between the available Rasuvo doses.
Rasuvo should not be used for the treatment of cancer.
Rasuvo should not be used for the treatment of children with psoriasis.
Description
Rasuvo contains methotrexate, a folate analog metabolic inhibitor.
Chemically, methotrexate is [N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]-Lglutamic acid. The structural formula is:
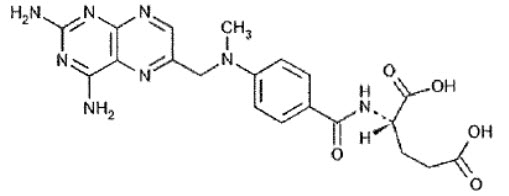
- C20H22N8O5
- M.W.= 454.45
Rasuvo contains methotrexate in a sterile, preservative-free, non-pyrogenic solution for a single subcutaneous injection. Rasuvo is an isotonic, clear, yellow to brown solution.
Rasuvo contains the following inactive ingredients: sodium chloride 0.4% w/v; water for injections, sodium hydroxide and, if necessary, hydrochloric acid are added to adjust the pH to approximately 8.5.
Mechanism of Action
Methotrexate inhibits dihydrofolic acid reductase. Dihydrofolates must be reduced to tetrahydrofolates by this enzyme before they can be utilized as carriers of one-carbon groups in the synthesis of purine nucleotides and thymidylate. Therefore, methotrexate interferes with DNA synthesis, repair, and cellular replication. Actively proliferating tissues such as malignant cells, bone marrow, fetal cells, buccal and intestinal mucosa, and cells of the urinary bladder are in general more sensitive to this effect of methotrexate.
The mechanism of action in rheumatoid arthritis is unknown; it may affect immune function.
What is the most important information I should know about Rasuvo?
Rasuvo can cause serious side effects that can lead to death, including:
1. Organ system toxicity. People who use methotrexate for the treatment of cancer, psoriasis, or rheumatoid arthritis, have an increased risk of death from organ toxicity. Types of organ toxicity can include:
- gastrointestinal
- bone marrow
- liver
- immune system
- nerve
- lung
- kidneys
- skin
Your doctor will do blood tests and other types of tests before you take and while you are taking Rasuvo to check for signs and symptoms of organ toxicity. Call your doctor right away if you have any of the following symptoms of organ toxicity:
- vomiting
- diarrhea
- mouth sores
- fever
- confusion
- weakness
- temporary blindness
- seizures
- headache
- back pain
- neck stiffness
- paralysis
- irritability
- sleepiness
- problems with coordination
- dry cough
- trouble breathing
- severe skin rash
- infection
2. Women who are pregnant are at increased risk for death of the baby and birth defects. Women who are pregnant or who plan to become pregnant must not take Rasuvo. A pregnancy test should be performed before starting Rasuvo.
Contraception should be used by both females and males while taking Rasuvo. Pregnancy should be avoided if either partner is receiving Rasuvo:
- for a minimum of 3 months after treatment with Rasuvo for males.
- during and for at least 1 menstrual cycle after treatment with Rasuvo for females.
Who should not take Rasuvo?
Do not take Rasuvo if you:
- are pregnant or planning to become pregnant. See “What is the most important information I should know about Rasuvo?”
- are breastfeeding.
- Rasuvo can pass into your breast milk and may harm your baby. Do not breastfeed while taking Rasuvo. Talk to your doctor about the best way to feed your baby if you take Rasuvo.
- have alcohol problems (alcoholism)
- have liver problems
- have problems fighting infection (immunodeficiency syndrome)
- have been told you have (or think you have) a blood disorder such as low levels of white blood cells, red blood cells (anemia), or platelets.
- have had an allergy to methotrexate or any of the ingredients in Rasuvo. See the end of this leaflet for a complete list of ingredients in Rasuvo.
Talk to your doctor before taking this medicine if you have any of these conditions.
What should I tell my healthcare provider before taking Rasuvo?
Before you take Rasuvo, tell your doctor if you have any other medical conditions.
Tell your doctor about all of the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements.
Rasuvo may affect how other medicines work, and other medicines may affect how Rasuvo works causing side effects.
Ask your doctor or pharmacist for a list of medicines if you are not sure.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take Rasuvo?
- Read the Instructions for Use that come with Rasuvo.
- Take Rasuvo exactly as your doctor tells you to take it.
- Inject Rasuvo only 1 time each week. Do not take Rasuvo every day.
- Taking Rasuvo every day may cause death from toxicity.
- Your doctor will show you or your caregiver how to inject Rasuvo. You should not inject Rasuvo until you have been trained on the right way to use it.
- Check Rasuvo before you inject it. Rasuvo should be yellow to brown in color and should not have any lumps or particles in it.
- Rasuvo should be injected under the skin of the abdomen or thigh.
- Do not inject Rasuvo within 2 inches of the belly button (navel)
- Use a different site each time you inject. This may help to decrease any reactions at the injection site.
- Do not inject Rasuvo in the arms or any other areas of the body.
- Do not inject Rasuvo in areas where the skin is tender, bruised, red, scaly, hard, or has scars or stretch marks.
- If you are not sure if Rasuvo was injected, or if you have hard time giving the injection, do not inject another dose. Call your pharmacist or doctor right away.
- If you inject too much Rasuvo, call your doctor or go to the nearest hospital emergency room right away.
What should I avoid while taking Rasuvo?
- Do not drink alcohol while taking Rasuvo. Drinking alcohol can increase your chances of getting serious side effects.
- Rasuvo can cause dizziness and tiredness. Do not drive a car, operate machinery, or do anything that needs you to be alert until you know how Rasuvo affects you.
- Certain vaccinations should be avoided while taking Rasuvo. Talk to your doctor before you or members of your household receive any vaccines.
What are the possible side effects of Rasuvo?
Rasuvo may cause serious side effects, including:
See “What is the most important information I should know about Rasuvo?”
- fertility problems. Methotrexate, the active ingredient in Rasuvo, may affect your ability to have a baby. Males may have a decreased sperm count, and females may have changes to their menstrual cycle. This can happen while taking Rasuvo and for a short period of time after you stop.
- certain cancers. Some people who have taken methotrexate have had a certain type of cancer called Non-Hodgkin’s lymphoma and other tumors. Your doctor may tell you to stop taking Rasuvo if this happens.
- tissue and bone problems. Taking methotrexate while having radiation therapy may increase the risk of your tissue or bone not receiving enough blood. This may lead to death of the tissue or bone.
Common side effects of Rasuvo include:
- nausea
- stomach pain
- indigestion (dyspepsia)
- mouth sores
- rash
- stuffy or runny nose and sore throat
- diarrhea
- abnormal liver function tests
- vomiting
- headache
- bronchitis
- low red, white, and platelet blood cell count
- hair loss
- dizziness
- sensitivity to light
- burning skin lesions
- lung problems
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Rasuvo. For more information, ask your doctor or pharmacist.
Call you doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Rasuvo
Methotrexate is sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use Rasuvo for a condition for which it was not prescribed. Do not give Rasuvo to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Rasuvo. If you would like more information, talk to your doctor. You can ask your doctor or pharmacist for information about Rasuvo that is written for health professionals.
For more information, please contact Medac Pharma, Inc. at our number 1-855-336-3322.
How should I store Rasuvo?
Store Rasuvo at room temperature between 68°F to 77°F (20°C to 25°C)
- Do not freeze
- Keep Rasuvo out of the light.
Keep Rasuvo and all medicines out of the reach of children.
How should I dispose of Rasuvo?
- Do not throw away in the household trash. Put used Rasuvo in an FDA-cleared sharps disposal container right away after use.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out
- upright stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about the safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Safely dispose of Rasuvo that is out of date or is no longer needed.
What are the ingredients in Rasuvo?
Active ingredient: methotrexate
Inactive ingredients: sodium chloride, sodium hydroxide and water for injection, USP, and if necessary hydrochloric acid, USP.
Label
PRINCIPAL DISPLAY PANEL – 22.5 MG AUTO-INJECTOR CARTON
- NDC 59137-535-04
Rx only - 22.5 mg
- Rasuvo®
methotrexate
injection - Rasuvo® (methotrexate) injection
- 22.5 mg per 0.45 mL
- This carton contains 4 pre-assembled 0.45 mL pre-filled Rasuvo auto-injectors.
FOR SUBCUTANEOUS USE ONLY
See full prescribing information for dosage and administration.
Read enclosed patient package insert carefully before using. 
PRINCIPAL DISPLAY PANEL – 25 MG AUTO-INJECTOR CARTON
- NDC 59137-540-04
Rx only - 25 mg
- Rasuvo®
methotrexate
injection - Rasuvo® (methotrexate) injection
- 25 mg per 0.50 mL
- This carton contains 4 pre-assembled 0.50 mL pre-filled Rasuvo auto-injectors.
FOR SUBCUTANEOUS USE ONLY
See full prescribing information for dosage and administration.
Read enclosed patient package insert carefully before using.
PRINCIPAL DISPLAY PANEL – 30 MG AUTO-INJECTOR CARTON
- NDC 59137-550-04
Rx only - 30 mg
- Rasuvo®
methotrexate
injection - Rasuvo® (methotrexate) injection
- 30 mg per 0.60 mL
- This carton contains 4 pre-assembled 0.60 mL pre-filled Rasuvo auto-injectors.
FOR SUBCUTANEOUS USE ONLY
See full prescribing information for dosage and administration.
Read enclosed patient package insert carefully before using.

SRC: NLM .
