Pralidoxime
Generic name: pralidoxime
Brand name: Protopam Chloride
Dosage form: intravenous powder for injection (1 g)
Drug class: Antidotes
Medically reviewed by A Ras MD.
What is pralidoxime?
Pralidoxime is a prescription medicine that is used to treat some overdoses. It is used to treat poisoning from nerve gas or chemicals that kill insects.
Description
Each prefilled ATNAA is a single-dose autoinjector that provides an intramuscular dose of atropine, a cholinergic muscarinic antagonist, and pralidoxime chloride, a cholinesterase reactivator, in a self-contained unit, specifically designed for automatic self- or buddy-administration by military personnel.
When activated, each ATNAA injection delivers the following:
- 2.1 mg of atropine in 0.7 mL of sterile, pyrogen-free solution containing 12.47 mg glycerin and not more than 2.8 mg phenol, citrate buffer, and Water for Injection. The pH range is 4.0 – 5.0.
- 600 mg of pralidoxime chloride equivalent to 476.6 mg of pralidoxime in 2 mL of sterile, pyrogen-free solution containing 40 mg benzyl alcohol, 22.5 mg glycine, and Water for Injection. The pH is adjusted with hydrochloric acid. The pH range is 2.0 – 3.0.
After ATNAA has been activated, the empty autoinjector should be disposed of properly. It cannot be refilled, nor can the protruding needle be retracted.
Atropine occurs as white crystals, usually needle-like, or as a white, crystalline powder. It is slightly soluble in water with a molecular weight of 289.38. Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and l-hyoscyamine, with activity due almost entirely to the levo isomer of the drug.
Chemically, atropine is designated as 1αH,5αH-Tropan-3α-ol (±)-tropate. Its empirical formula is C17H23NO3 and its structural formula is as follows:
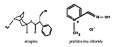
Pralidoxime chloride is an odorless, white to pale-yellow crystalline powder, freely soluble in water, with a molecular weight of 172.61. Chemically, pralidoxime chloride is designated as 2-formyl-1-methylpyridinium chloride oxime. Its empirical formula is C7H9CIN2O and its structural formula is indicated above.
Mechanism of Action
Atropine
Atropine competitively blocks the effects of acetylcholine, including excess acetylcholine due to organophosphorus nerve agent poisoning, at muscarinic cholinergic receptors on smooth muscle, cardiac muscle, secretory gland cells, and in peripheral autonomic ganglia and the central nervous system.
Pralidoxime Chloride
Pralidoxime chloride reactivates acetylcholinesterase which has been inactivated by phosphorylation due to susceptible organophosphorus nerve agents having anticholinesterase activity . However, pralidoxime chloride does not reactivate acetylcholinesterase inactivated by all organophosphorus nerve agents (e.g. soman). Pralidoxime chloride cannot reactivate phosphorylated acetylcholinesterases that have undergone a further chemical reaction known as “aging.” Reactivated acetylcholinesterase hydrolyzes excess acetylcholine resulting from organophosphorus nerve agent poisoning to help restore impaired cholinergic neural function. Reactivation is clinically important because only a small proportion of active acetylcholinesterase is needed to maintain vital functions.
Before taking pralidoxime, tell your doctor:
- If you are allergic to pralidoxime; any part of this medicine; or any other drugs, foods, or substances. Tell your doctor about the allergy and what signs you had.
This medicine may interact with other drugs or health problems.
Tell your doctor and pharmacist about all of your drugs (prescription or OTC, natural products, vitamins) and health problems. You must check to make sure that it is safe for you to take pralidoxime with all of your drugs and health problems. Do not start, stop, or change the dose of any drug without checking with your doctor.
What are some things I need to know or do while I take pralidoxime?
- Tell all of your health care providers that you take pralidoxime. This includes your doctors, nurses, pharmacists, and dentists.
- Have blood work checked as you have been told by the doctor. Talk with the doctor.
- Tell your doctor if you are pregnant or plan on getting pregnant. You will need to talk about the benefits and risks of using pralidoxime while you are pregnant.
- Tell your doctor if you are breast-feeding. You will need to talk about any risks to your baby.
How is pralidoxime best taken?
Use pralidoxime as ordered by your doctor. Read all information given to you. Follow all instructions closely.
- It is given into a vein for a period of time.
- It may be given as a shot into a muscle.
- It may be given as a shot into the fatty part of the skin.
What are the side effects of pralidoxime that I need to call my doctor about immediately?
WARNING/CAUTION: Even though it may be rare, some people may have very bad and sometimes deadly side effects when taking a drug. Tell your doctor or get medical help right away if you have any of the following signs or symptoms that may be related to a very bad side effect:
- Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing, swallowing, or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat.
- Signs of high blood pressure like very bad headache or dizziness, passing out, or change in eyesight.
- A fast heartbeat.
- Fast breathing.
- Change in eyesight.
- Muscle weakness.
- Signs that are worse have happened when pralidoxime was given into a vein very fast. This includes chest pain or pressure, fast heartbeat, throat irritation, rigid muscles, or not being able to move. If any of these side effects happen, tell the doctor right away.
What are some other side effects of pralidoxime?
All drugs may cause side effects. However, many people have no side effects or only have minor side effects. Call your doctor or get medical help if any of these side effects or any other side effects bother you or do not go away:
Injection:
- Headache.
- Dizziness.
- nausea
- Feeling sleepy.
- Upset stomach.
Injection (if given in the muscle):
- Pain where the shot was given.
These are not all of the side effects that may occur. If you have questions about side effects, call your doctor. Call your doctor for medical advice about side effects.
You may report side effects to the FDA at 1-800-332-1088. You may also report side effects at https://www.fda.gov/medwatch.
If overdose is suspected:
If you think there has been an overdose, call your poison control center or get medical care right away. Be ready to tell or show what was taken, how much, and when it happened.
How do I store and/or throw out pralidoxime?
- If you need to store pralidoxime at home, talk with your doctor, nurse, or pharmacist about how to store it.
Label
PRINCIPAL DISPLAY PANEL
- Principal Display Panel
- ANTIDOTE TREATMENT NERVE AGENT, AUTO-INJECTOR
- For Use in Nerve Agent Poisoning Only
- ATROPINE INJECTION 2.1 MG,
- PRALIDOXIME CHLORIDE INJECTION 600MG
- MERIDIAN MEDICAL TECHNOLOGIES®
- Columbia, MD 21046, USA
- A Pfizer Company.

- ATNAA Utilizes Binaject®Technology 0001860
- Sterile Solution for Intramuscular Use Only
- NSN 6505-01-362-7427
- NDC 11704-777-01
- Rx Only
- Store at 25°C (77°F); excursions permitted to 15 – 30°C (59-86°F).
- Keep From Freezing. Protect From Light.

SRC: NLM .
