Pemfexy
Generic name: pemetrexed (intravenous route)
Drug class: Antimetabolites
Medically reviewed by A Ras MD.
What is Pemfexy?
Pemfexy is a prescription medicine used to treat a kind of lung cancer called non-squamous non-small cell lung cancer (NSCLC). Pemfexy is also used:
-
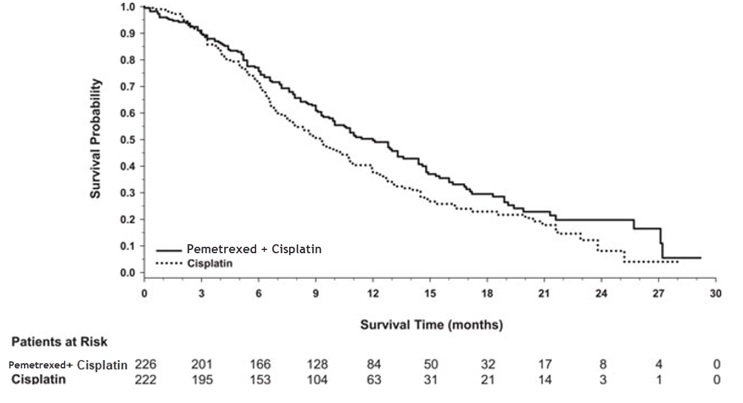
- as the first treatment in combination with cisplatin when your lung cancer has spread (advanced NSCLC).
- alone as maintenance treatment after you have received 4 cycles of chemotherapy that contains platinum for first treatment of your advanced NSCLC and your cancer has not progressed.
- alone when your lung cancer has returned or spread after prior chemotherapy.
Pemfexy is not for use for the treatment of people with squamous cell non-small cell lung cancer.
- a kind of cancer called malignant pleural mesothelioma. This cancer affects the lining of the lungs and chest wall. Pemfexy is used in combination with cisplatin as the first treatment for malignant pleural mesothelioma that cannot be removed by surgery or you are not able to have surgery.
It is not known if Pemfexy is safe and effective in children.
Description
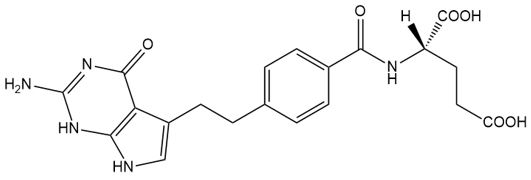
Pemetrexed is a folate analog metabolic inhibitor. Pemetrexed diacid, the drug substance, has the chemical name N-[4-[2-(2-amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]-L-glumatic acid. The molecular formula is C20H21N5O6 and the molecular weight is 427.41. The structural formula is as follows:
PEMFEXY (pemetrexed injection) for intravenous use is a sterile, clear, colorless to yellow or green-yellow solution . Each mL contains: 25 mg pemetrexed diacid, 260 mg propylene glycol, up to 16.5-19.9 mg tromethamine, and water for injection. Additional tromethamine not exceeding 19.9 mg/mL and/or hydrochloric acid may be added for pH adjustment.
Mechanism of Action
Pemetrexed is a folate analog metabolic inhibitor that disrupts folate-dependent metabolic processes essential for cell replication. In vitro studies show that pemetrexed inhibits thymidylate synthase (TS), dihydrofolate reductase (DHFR), and glycinamide ribonucleotide formyltransferase (GARFT), which are folate-dependent enzymes involved in the de novo biosynthesis of thymidine and purine nucleotides. Pemetrexed is taken into cells by membrane carriers, such as the reduced folate carrier and membrane folate binding protein transport systems. Once in the cell, pemetrexed is converted to polyglutamate forms by the enzyme folylpolyglutamate synthetase. The polyglutamate forms are retained in cells and are inhibitors of TS and GARFT.
Who should not use Pemfexy?
Do not take Pemfexy if you have had a severe allergic reaction to any medicine that contains pemetrexed.
What should I tell my healthcare provider before using Pemfexy?
Before taking Pemfexy, tell your healthcare provider about all of your medical conditions, including if you:
- have kidney problems.
- have had radiation therapy.
- are pregnant or plan to become pregnant. Pemfexy can harm your unborn baby.
- Your healthcare provider should do a pregnancy test before you start your treatment with Pemfexy.
- Females who are able to become pregnant should use effective birth control (contraception) during treatment with Pemfexy and for 6 months after the final dose. Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with Pemfexy.
- Males with female partners who are able to become pregnant should use effective birth control (contraception) during treatment with Pemfexy and for 3 months after the final dose.
- are breastfeeding or plan to breastfeed. It is not known if Pemfexy passes into breast milk. Do not breastfeed during treatment with Pemfexy and for 1 week after the final dose.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Tell your healthcare provider if you have kidney problems and take a medicine that contains ibuprofen. You should avoid taking ibuprofen for 2 days before, the day of, and 2 days after receiving treatment with Pemfexy.
How should I use Pemfexy?
- It is very important to take folic acid and vitamin B12 during your treatment with Pemfexy to lower your risk of harmful side effects.
- Take folic acid exactly as prescribed by your healthcare provider 1 time a day, beginning 7 days (1 week) before your first dose of Pemfexy and continue taking folic acid until 21 days (3 weeks) after your last dose of Pemfexy.
- Your healthcare provider will give you vitamin B12 injections during treatment with Pemfexy. You will get your first vitamin B12 injection 7 days (1 week) before your first dose of Pemfexy, and then every 3 cycles.
- Your healthcare provider will prescribe a medicine called corticosteroid for you to take 2 times a day for 3 days, beginning the day before each treatment with Pemfexy.
- Pemfexy is given to you by intravenous (IV) infusion into your vein. The infusion is given over 10 minutes.
- Pemfexy is usually given 1 time every 21 days (3 weeks).
What are the possible side effects of Pemfexy?
Pemfexy can cause serious side effects, including:
- Low blood cell counts. Low blood cell counts can be severe, including low white blood cell counts (neutropenia), low platelet counts (thrombocytopenia), and low red blood cell counts (anemia). Your healthcare provider will do blood test to check your blood cell counts regularly during your treatment with Pemfexy. Tell your healthcare provider right away if you have any signs of infection, fever, bleeding, or severe tiredness during your treatment with Pemfexy.
- Kidney problems, including kidney failure. Pemfexy can cause severe kidney problems that can lead to death. Severe vomiting or diarrhea can lead to loss of fluids (dehydration) which may cause kidney problems to become worse. Tell your healthcare provider right away if you have a decrease in the amount of urine you make.
- Severe skin reactions. Severe skin reactions that may lead to death can happen with Pemfexy. Tell your healthcare provider right away if you develop blisters, skin sores, skin peeling, or painful sores, or ulcers in your mouth, nose, throat or genital area.
- Lung problems (pneumonitis). Pemfexy can cause serious lung problems that can lead to death. Tell your healthcare provider right away if you get any new or worsening symptoms of shortness of breath, cough, or fever.
- Radiation recall. Radiation recall is a skin reaction that can happen in people who have received radiation treatment in the past and are treated with Pemfexy. Tell your healthcare provider if you get swelling, blistering, or a rash that looks like a sunburn in an area that was previously treated with radiation.
The most common side effects of Pemfexy when given alone are:
- tiredness
- nausea
- loss of appetite
The most common side effects of Pemfexy when given with cisplatin are:
- vomiting
- low white blood cell counts (neutropenia)
- swelling or sores in your mouth or sore throat
- low platelet counts (thrombocytopenia)
- constipation
- low red blood cell counts (anemia)
Pemfexy may cause fertility problems in males. This may affect your ability to father a child. It is not known if these effects are reversible. Talk to your healthcare provider if this is a concern for you.
Your healthcare provider will do blood test to check for side effects during treatment with Pemfexy. Your healthcare provider may change your dose of Pemfexy, delay treatment, or stop treatment if you have certain side effects.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of Pemfexy. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Pemfexy
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information guide. You can ask your pharmacist or healthcare provider for information about Pemfexy that is written for health professionals.
What are the ingredients in Pemfexy?
Active ingredient: pemetrexed
Inactive ingredients: propylene glycol, tromethamine, and water for injection. Additional tromethamine or hydrochloric acid may be added for pH adjustment.
Label
PRINCIPAL DISPLAY PANEL – NDC: 42367-531-33 – 25MG/ML VIAL LABEL
PRINCIPAL DISPLAY PANEL – NDC: 42367-531-33 – 25MG/ML CARTON LABEL

