Onureg
Generic name: azacitidine
Drug class: Miscellaneous antineoplastics
Medically reviewed by A Ras MD.
What is Onureg?
Onureg is a prescription medicine used for continued treatment of adults with acute myeloid leukemia (AML) who had a first complete remission (CR) following intensive induction chemotherapy with or without recovery of your blood cell counts, and who are not able to complete intensive curative therapy.
It is not known if Onureg is safe and effective in children under 18 years of age.
Description
Azacitidine is a nucleoside metabolic inhibitor with a molecular formula of C8H12N4O5 and a molecular weight of 244 g/mol. The chemical name is: 4-amino-1-β-D-ribofuranosyl-s-triazin-2(1H)-one and the chemical structural is:

Azacitidine is a white to off-white solid. Azacitidine was found to be soluble in aqueous media across a pH range from 1.0 to 7.0.
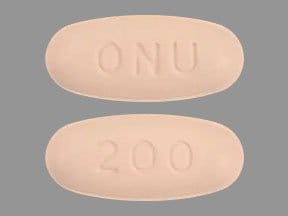
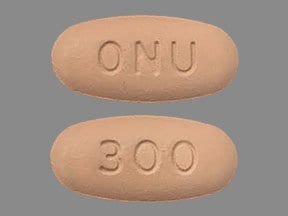
ONUREG (azacitidine) is supplied as film-coated tablets containing 200 mg or 300 mg of azacitidine for oral use. Each core tablet contains the following inactive ingredients: croscarmellose sodium, magnesium stearate, mannitol, and silicified microcrystalline cellulose. The 200 and 300 mg tablet coating contains hypromellose, lactose monohydrate, polyethylene glycol, titanium dioxide, and triacetin. In addition, the 200 mg tablet coating contains iron oxide red and the 300 mg tablet coating contains black iron oxide, iron oxide red, and iron oxide yellow.
Mechanism of Action
Azacitidine is a pyrimidine nucleoside analog of cytidine that inhibits DNA/RNA methyltransferases. Azacitidine is incorporated into DNA and RNA following cellular uptake and enzymatic biotransformation to nucleotide triphosphates.
Incorporation of azacitidine into the DNA of cancer cells in vitro, including acute myeloid leukemia cells, inhibited DNA methyltransferases, reduced DNA methylation and altered gene expression, including re-expression of genes regulating tumor suppression and cell differentiation. Incorporation of azacitidine into the RNA of cancer cells, including leukemic cells, inhibited RNA methyltransferases, reduced RNA methylation, decreased RNA stability and decreased protein synthesis.
Antileukemic activity of azacitidine was demonstrated by reduction of cell viability and induction of apoptosis in AML cell lines in vitro. Azacitidine decreased tumor burden and increased survival in leukemic tumor models in vivo.
Who should not take Onureg?
Do not take Onureg if you are allergic to azacitidine or any of the ingredients in Onureg. See the end of this guide for a complete list of ingredients in Onureg.
What should I tell my healthcare provider before taking Onureg?
Before taking Onureg, tell your healthcare provider about all of your medical conditions, including if you:
- have kidney or liver problems.
- are pregnant or plan to become pregnant. Onureg can harm your unborn baby.
Females who are able to become pregnant:- Your healthcare provider should perform a pregnancy test before you start treatment with Onureg.
- You should use effective birth control (contraception) during treatment and for at least 6 months after your last dose of Onureg.
- Tell your healthcare provider right away if you become pregnant during treatment with Onureg.
Males with a female sexual partner who can become pregnant:
- You should use effective birth control (contraception) during treatment and for at least 3 months after your last dose of Onureg.
- are breastfeeding or plan to breastfeed. It is not known if Onureg passes into your breast milk. Do not breastfeed during treatment and for 1 week after your last dose of Onureg.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I take Onureg?
- Take Onureg exactly as your healthcare provider tells you to take it.
- Your healthcare provider will prescribe an anti-nausea medicine for you to take to help prevent nausea and vomiting during your treatment with Onureg.
- Take the anti-nausea medicine 30 minutes before each dose of Onureg.
- Your healthcare provider may decide to stop the anti-nausea medicine after your second cycle of Onureg, if you do not have any nausea or vomiting.
- Take Onureg by mouth 1 time each day beginning on Day 1 through Day 14 of each 28-day cycle.
- Take Onureg with or without food at about the same time each day.
- Swallow Onureg tablets whole. Do not cut, split, crush, or chew the tablets.
- If the powder from Onureg tablets comes in contact with your skin, wash the area well right away with soap and water.
- If the powder from Onureg tablets comes in contact with your eyes or mouth (mucous membranes), flush the area right away with water.
- If you miss a dose of Onureg, or if you do not take your dose at the usual time, take the dose as soon as possible that day. Take your next dose at the regular time the next day. Do not take 2 doses on the same day to make up for a missed dose.
- If you vomit after taking a dose of Onureg, do not take another dose on the same day. Take your next dose at the regular time the next day.
What are the possible side effects of Onureg?
Onureg can cause serious side effects, including:
- New or worsening low white blood cell counts (neutropenia). New or worsening low white blood cell counts are common but can also be severe during treatment with Onureg. If your white blood cell counts become very low, you are at increased risk for infections. Your healthcare provider will check your white blood cell counts before and during treatment with Onureg. Your healthcare provider may prescribe a medicine to help increase your white blood cell count if needed.
Tell your healthcare provider right away if you get any of the following symptoms:- fever or chills
- body aches
- feeling very tired or weak
- unusual headaches
- New or worsening low platelet counts (thrombocytopenia). Low platelet counts are common but can also be severe during treatment with Onureg. Your healthcare provider will check your platelet counts before and during treatment with Onureg. Tell your healthcare provider right away if you have any unusual bruising or bleeding. Your healthcare provider may change your dose or tell you to stop taking Onureg if you have low blood cell counts.
Onureg may cause fertility problems in males and females, which may affect your ability to have children. Talk with your healthcare provider if you have concerns about fertility.
The most common side effects of Onureg include:
- nausea and vomiting. See “How should I take Onureg?”
- diarrhea. You may need to be treated with anti-diarrheal medicines.
- tiredness or weakness
- constipation
- stomach area (abdominal) pain
- pneumonia
- joint pain
- decreased appetite
- pain in arms or legs
- dizziness
These are not all of the possible side effects of Onureg. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Onureg
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information guide. Do not use Onureg for a condition for which it was not prescribed. Do not give Onureg to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Onureg that is written for health professionals.
How should I store Onureg?
- Store bottles of Onureg tablets at room temperature between 68°F to 77°F (20°C to 25°C).
- Store Onureg tablets in the original bottle.
- Bottles of Onureg contain 2 drying agent (desiccant) canisters. Do not eat the desiccant canisters.
- Keep the Onureg bottle tightly closed.
- Talk to your healthcare provider about how to safely throw away (dispose of) any unused or expired Onureg.
Keep Onureg and all medicines out of the reach of children
What are the ingredients in Onureg?
Active ingredient: azacitidine
Inactive ingredients:
Each core tablet contains: croscarmellose sodium, magnesium stearate, mannitol, and silicified microcrystalline cellulose.
The pink 200 mg tablet coating contains: hypromellose, iron oxide red, lactose monohydrate, polyethylene glycol, titanium dioxide, and triacetin.
The brown 300 mg tablet coating contains: black iron oxide, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, polyethylene glycol, titanium dioxide, and triacetin.
Label
PRINCIPAL DISPLAY PANEL – 200 MG BLISTER PACK CARTON
- NDC 59572-730-07
Rx only - ONUREG™
(azacitidine) tablets - 200 mg
- Each tablet contains 200 mg of azacitidine.
- Swallow tablets whole. Do not cut, crush, or chew the tablets.
- One Blister Card
Containing 7 Tablets - Celgene
- CAUTION: Hazardous Agent

PRINCIPAL DISPLAY PANEL – 300 MG BOTTLE LABEL
- NDC 59572-740-14
- ONUREG™
(azacitidine) tablets - 300 mg
- Swallow tablets whole.
Do not cut, crush, or chew the tablets. - Rx only
14 Tablets - CAUTION: Hazardous Agent

SRC: NLM .