Nesina
Generic name: alogliptin
Drug class: Dipeptidyl peptidase 4 inhibitors
Medically reviewed by A Ras MD. Last updated on May 23, 2022.
What is Nesina?
Nesina is a prescription medicine used along with diet and exercise to improve blood sugar (glucose) control in adults with type 2 diabetes.
Nesina is not for people with type 1 diabetes.
It is not known if Nesina is safe and effective in children under the age of 18.
Description:
NESINA tablets contain the active ingredient alogliptin, which is a selective, orally bioavailable inhibitor of the enzymatic activity of dipeptidyl peptidase-4 (DPP-4).
Chemically, alogliptin is prepared as a benzoate salt, which is identified as 2-({6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl}methyl)benzonitrile monobenzoate. It has a molecular formula of C18H21N5O2•C7H6O2 and a molecular weight of 461.51 daltons. The structural formula is:
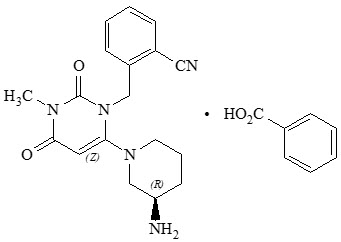
Alogliptin benzoate is a white to off-white crystalline powder containing one asymmetric carbon in the aminopiperidine moiety. It is soluble in dimethylsulfoxide, sparingly soluble in water and methanol, slightly soluble in ethanol and very slightly soluble in octanol and isopropyl acetate.
Each NESINA tablet contains 34 mg, 17 mg or 8.5 mg alogliptin benzoate, which is equivalent to 25 mg, 12.5 mg or 6.25 mg, respectively, of alogliptin and the following inactive ingredients: mannitol, microcrystalline cellulose, hydroxypropyl cellulose, croscarmellose sodium and magnesium stearate. In addition, the film coating contains the following inactive ingredients: hypromellose, titanium dioxide, ferric oxide (red or yellow) and polyethylene glycol, and is marked with printing ink (Gray F1).
Mechanism of Action
Increased concentrations of the incretin hormones such as glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are released into the bloodstream from the small intestine in response to meals. These hormones cause insulin release from the pancreatic beta cells in a glucose-dependent manner but are inactivated by the dipeptidyl peptidase-4 (DPP-4) enzyme within minutes. GLP-1 also lowers glucagon secretion from pancreatic alpha cells, reducing hepatic glucose production.
In patients with type 2 diabetes, concentrations of GLP-1 are reduced but the insulin response to GLP-1 is preserved. Alogliptin is a DPP-4 inhibitor that slows the inactivation of the incretin hormones, thereby increasing their bloodstream concentrations and reducing fasting and postprandial glucose concentrations in a glucose-dependent manner in patients with type 2 diabetes mellitus. Alogliptin selectively binds to and inhibits DPP-4 but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
What is the most important information I should know about Nesina?
Serious side effects can happen to people taking Nesina, including:
1. Inflammation of the pancreas (pancreatitis): Nesina may cause pancreatitis which may be severe. Certain medical conditions make you more likely to get pancreatitis.
Before you start taking Nesina:
Tell your doctor if you have ever had:
- pancreatitis
- kidney problems
- liver problems
- high blood triglyceride levels
- stones in your gallbladder (gall stones)
- a history of alcoholism
Stop taking Nesina and call your doctor right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis.
2. Heart failure: Heart failure means your heart does not pump blood well enough.
Before you start taking Nesina:
Tell your doctor if you have ever had heart failure or have problems with your kidneys.
Contact your doctor right away if you have any of the following symptoms:
- increasing shortness of breath or trouble breathing especially when lying down
- an unusually fast increase in weight
- swelling of feet, ankles, or legs
- unusual tiredness
These may be symptoms of heart failure.
Who should not take Nesina?
Do not take Nesina if you:
- Are allergic to any ingredients in Nesina or have had a serious allergic (hypersensitivity) reaction to Nesina. See the end of this Medication Guide for a complete list of the ingredients in Nesina. Symptoms of a serious allergic reaction to Nesina may include:
- swelling of your face, lips, throat and other areas on your skin
- difficulty with swallowing or breathing
- raised, red areas on your skin (hives)
- skin rash, itching, flaking or peeling
If you have any of these symptoms, stop taking Nesina and contact your doctor or go to the nearest hospital emergency room right away.
What should I tell my healthcare provider before taking Nesina?
Before you take Nesina, tell your doctor if you:
- have heart failure
- have or have had inflammation of your pancreas (pancreatitis)
- have kidney or liver problems
- have other medical conditions
- are pregnant or plan to become pregnant. It is not known if Nesina can harm your unborn baby. Talk with your doctor about the best way to control your blood sugar while you are pregnant or if you plan to become pregnant
- are breastfeeding or plan to breastfeed. It is not known if Nesina passes into your breast milk. Talk with your doctor about the best way to feed your baby if you are taking Nesina
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist before you start any new medicine.
Nesina may affect the way other medicines work, and other medicines may affect how Nesina works. Contact your doctor before you start or stop other types of medicines.
How should I take Nesina?
- Take Nesina exactly as your doctor tells you to take it.
- Take Nesina 1 time each day with or without food.
- Do not split tablets.
- If you miss a dose, take it as soon as you remember. If you do not remember until it is time for your next dose, skip the missed dose, and take the next dose at your regular time. Do not take 2 doses of Nesina at the same time.
- If you take too much Nesina, call your doctor or your poison control center right away at 1-800-222-1222.
- If your body is under stress, such as from fever, infection, accident or surgery, the dose of your diabetes medicines may need to be changed. Call your doctor right away.
- Stay on your diet and exercise programs and check your blood sugar as your doctor tells you to.
- Your doctor may do certain blood tests before you start Nesina and during treatment as needed. Your doctor may change your dose of Nesina based on the results of your blood tests due to how well your kidneys are working.
- Your doctor will check your diabetes with regular blood tests, including your blood sugar levels and your hemoglobin A1C.
What are the possible side effects of Nesina?
Nesina can cause serious side effects, including:
See “What is the most important information I should know about Nesina?”
- Serious allergic (hypersensitivity) reactions such as:
- swelling of your face, lips, throat and other areas on your skin
- difficulty swallowing or breathing
- raised, red areas on your skin (hives)
- skin rash, itching, flaking or peeling If you have these symptoms, stop taking Nesina and contact your doctor right away or go the nearest hospital emergency room.
- Liver problems. Call your doctor right away or go to the nearest hospital emergency room if you have unexplained symptoms, such as:
- nausea or vomiting
- stomach pain
- unusual or unexplained tiredness
- loss of appetite
- dark urine
- yellowing of your skin or the whites of your eye
- Low blood sugar (hypoglycemia). If you take Nesina with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin, your risk of getting low blood sugar is higher. The dose of your sulfonylurea medicine or insulin may need to be lowered while you take Nesina. If you have symptoms of low blood sugar, you should check your blood sugar and treat if low, then call your doctor. Signs and symptoms of low blood sugar include:
- shaking or feeling jittery
- sweating
- hunger
- headache
- change in mood
- fast heartbeat
- change in vision
- confusion
- dizziness
- Joint pain. Some people who take medicines called DPP-4 inhibitors like Nesina, may develop joint pain that can be severe. Call your doctor if you have severe joint pain.
- Skin reaction. Some people who take medicines called DPP-4 inhibitors, like Nesina, may develop a skin reaction called bullous pemphigoid that can require treatment in a hospital. Tell your doctor right away if you develop blisters or the breakdown of the outer layer of your skin (erosion). Your doctor may tell you to stop taking Nesina.
The most common side effects of Nesina include stuffy or runny nose and sore throat, headache, or cold-like symptoms (upper respiratory tract infection).
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Nesina. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Label:
NESINA tablets are available as film-coated tablets containing 25 mg, 12.5 mg or 6.25 mg of alogliptin as follows:
25 mg tablet: light red, oval, biconvex, film-coated, with “TAK ALG-25” printed on one side, available in:
| NDC 64764-250-30 | Bottles of 30 tablets |
| NDC 64764-250-90 | Bottles of 90 tablets |
| NDC 64764-250-50 | Bottles of 500 tablets |
12.5 mg tablet: yellow, oval, biconvex, film-coated, with “TAK ALG-12.5” printed on one side, available in:
| NDC 64764-125-30 | Bottles of 30 tablets |
| NDC 64764-125-90 | Bottles of 90 tablets |
| NDC 64764-125-50 | Bottles of 500 tablets |
6.25 mg tablet: light pink, oval, biconvex, film-coated, with “TAK ALG-6.25” printed on one side, available in:
| NDC 64764-625-30 | Bottles of 30 tablets |
| NDC 64764-625-90 | Bottles of 90 tablets |
Storage
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
PRINCIPAL DISPLAY PANEL – 6.25 MG TABLET BOTTLE LABEL
- NDC 64764-625-30
- Nesina
(alogliptin) tablets - 6.25 mg
- Dispense with
Medication Guide - 30 Tablets
- Takeda
- Rx Only

PRINCIPAL DISPLAY PANEL – 12.5 MG TABLET BOTTLE LABEL
- NDC 64764-125-30
- Nesina
(alogliptin) tablets - 12.5 mg
- Dispense with
Medication Guide - 30 Tablets
- Takeda
- Rx Only

PRINCIPAL DISPLAY PANEL – 25 MG TABLET BOTTLE LABEL
- NDC 64764-250-30
- Nesina
(alogliptin) tablets - 25 mg
- Dispense with
Medication Guide - 30 Tablets
- Takeda
- Rx Only

General information about the safe and effective use of Nesina
Medicines are sometimes prescribed for purposes other than those listed in the Medication Guide. Do not take Nesina for a condition for which it was not prescribed. Do not give Nesina to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about Nesina. If you would like to know more information, talk with your doctor. You can ask your doctor or pharmacist for information about Nesina that is written for health professionals.
For more information go to www.NESINA.com or call 1-877-TAKEDA-7 (1-877-825-3327).
How should I store Nesina?
Store Nesina at room temperature between 68°F to 77°F (20°C to 25°C).
Keep Nesina and all medicines out of the reach of children.
What are the ingredients in Nesina?
Active ingredient: alogliptin
Inactive ingredients: mannitol, microcrystalline cellulose, hydroxypropyl cellulose, croscarmellose sodium and magnesium stearate. In addition, the film-coating contains the following inactive ingredients: hypromellose, titanium dioxide, ferric oxide (red or yellow) and polyethylene glycol and is marked with gray F1 printing ink
SRC: NLM .



