Myalept
Generic name: metreleptin
Drug class: Miscellaneous metabolic agents
Medically reviewed by A Ras MD.
What is Myalept?
Myalept is a prescription medicine used with a diet recommended by your healthcare provider to treat problems caused by not having enough leptin in your body (leptin deficiency) in people with congenital or acquired generalized lipodystrophy.
- It is not known if Myalept is safe and effective when used:
- to treat problems (complications) caused by partial lipodystrophy
- to treat liver disease, including non-alcoholic steatohepatitis (NASH)
- Myalept should not be used to treat:
- people with HIV-related lipodystrophy
- people with metabolic disease, including diabetes mellitus and hypertriglyceridemia, without signs or symptoms of congenital or acquired generalized lipodystrophy
Description
MYALEPT (metreleptin) for injection is a recombinant human leptin analog for injection that binds to and activates the leptin receptor. Metreleptin (recombinant methionyl-human leptin) is produced in E. coli and differs from native human leptin by the addition of a methionine residue at its amino terminus. Metreleptin is a 147-amino acid, nonglycosylated, polypeptide with one disulfide bond between Cys-97 and Cys-147 and a molecular weight of approximately 16.15 kDa.
MYALEPT is supplied as a sterile, white, solid, lyophilized cake containing 11.3 mg that is reconstituted with 2.2 mL of BWFI or WFI to a final formulation of 5 mg/mL metreleptin for subcutaneous injection. Inactive ingredients are: glutamic acid (1.47 mg/mL), glycine (20 mg/mL), polysorbate 20 (0.1 mg/mL), and sucrose (10 mg/mL), pH 4.25.
Mechanism of Action
Adipocytes store lipids to meet the fuel requirements of non-adipose tissues during fasting. In patients with generalized lipodystrophy, the deficiency of adipose tissue leads to hypertriglyceridemia and ectopic deposition of fat in non-adipose tissues such as liver and muscle, contributing to metabolic abnormalities including insulin resistance. Native leptin is a hormone predominantly secreted by adipose tissue that informs the central nervous system of the status of energy stores in the body. In patients with generalized lipodystrophy, leptin deficiency, resulting from the loss of adipose tissue, contributes to excess caloric intake, which exacerbates the metabolic abnormalities.
MYALEPT (metreleptin) for injection exerts its function by binding to and activating the human leptin receptor (ObR), which belongs to the Class I cytokine family of receptors that signals through the JAK/STAT transduction pathway.
What is the most important information I should know about Myalept?
Myalept may cause serious side effects, including:
- risk for loss of endogenous leptin activity or loss of Myalept efficacy due to neutralizing antibodies.
Some people who use Myalept make antibodies in their blood that may reduce how well the leptin in your body (endogenous) works or how well Myalept works. Side effects may include:- infection
- problems with blood sugar, including diabetes
- an increase in the amount of fat in your blood (triglycerides)
- lymphoma (a type of blood cancer). There may be an increased risk of getting lymphoma when you use Myalept.
Myalept is only available through a restricted program called the Myalept Risk Evaluation and Mitigation Strategy (REMS) Program. For more information about the Myalept REMS Program go to www.myaleptrems.com or call 1-855-669-2537.
Who should not use Myalept?
Do not use Myalept if you:
- have general obesity that is not caused by a congenital leptin deficiency.
- are allergic to metreleptin or any of the ingredients in Myalept. See the end of this Medication Guide for a complete list of ingredients in Myalept.
Talk to your healthcare provider right away if you have any symptoms of an allergic reaction including a rash or itching (hives).
Symptoms of a severe allergic reaction may include:- swelling of your face, lips, tongue, or throat
- problems breathing or swallowing
- severe rash or itching
- fainting or feeling dizzy
- very rapid heartbeat
What should I tell my healthcare provider before using Myalept?
Before using Myalept, tell your healthcare provider if you:
- have or have had problems with your blood cells including low blood cell counts, especially white blood cells
- have or have had problems with your bone marrow
- have or have had swollen lymph nodes (lymphadenopathy)
- have or have had lymphoma
- use insulin or a sulfonylurea
- have or have had problems with your immune system (autoimmune disease)
- have or have had problems with your pancreas (pancreatitis)
- have high blood triglyceride levels
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Myalept will harm your unborn baby
- If you become pregnant while using Myalept, talk to your healthcare provider about registering with a program to collect information about the outcomes of moms and babies exposed to Myalept during pregnancy. You can enroll in the Myalept program by calling 1-855-669-2537.
- are breastfeeding or plan to breastfeed. It is not known if Myalept passes into your breast milk. You and your healthcare provider should decide if you will take Myalept or breastfeed. You should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Myalept?
- See the Instructions for Use that come with Myalept for detailed instructions on using Myalept the right way.
- Use Myalept exactly as your healthcare provider tells you to.
- Your healthcare provider will tell you how much Myalept to use and when to use it. Do not change your dose unless your healthcare provider tells you to.
- Do not suddenly stop using Myalept. Stopping Myalept suddenly may cause a serious problem with your pancreas (pancreatitis) and very high triglycerides.
- If your healthcare provider decides that you should stop using Myalept, your healthcare provider should slowly decrease your dose (taper) over 1 week.
- Myalept is injected 1 time a day at the same time each day.
- Myalept can be used with or without food.
- If you miss a dose of Myalept, take it as soon as you remember. Take your regular dose the next day at your normal time. Do not take more than your regular daily dose in a single day. Do not take an extra dose or increase the amount of your dose to make up for a missed dose.
- Do not mix Myalept and insulin in the same syringe or vial. Although Myalept and insulin doses may be given at the same time, do not inject Myalept and insulin in the same injection site.
- When Myalept is used in newborns or infants, your healthcare provider will tell you if Myalept should be mixed with a liquid called sterile water for injection (WFI) (preservative-free). For older children and adults, bacteriostatic water for injection (BWFI) may be used.
- Myalept is given as an injection under the skin (subcutaneous) of your stomach (abdomen), thigh, or upper arm. Do not inject Myalept into a vein or muscle.
What are the possible side effects of Myalept?
Myalept can cause serious side effects, including:
- See “What is the most important information I should know about Myalept?”
- low blood sugar (hypoglycemia). You may get low blood sugar if you use Myalept with another medicine that can cause low blood sugar, such as insulin or sulfonylurea. The dose of your insulin or sulfonylurea may need to be lowered while you use Myalept. Signs and symptoms of low blood sugar may include:
- shakiness
- sweating
- headache
- drowsiness
- weakness
- dizziness
- confusion
- irritability
- hunger
- fast heart beat
- feeling jitteryTalk with your healthcare provider about how to recognize and treat low blood sugar. Make sure that your family and other people around you a lot know how to recognize and treat low blood sugar.
- autoimmunity. People who have or have had certain problems with their immune system (autoimmune disease) may have worsening of their symptoms with Myalept. Talk to your healthcare provider about what symptoms you should watch for that would warrant further testing.
- allergic reactions (hypersensitivity). Allergic reactions can happen in people who use Myalept. Talk to your healthcare provider right away if you have any symptom of an allergic reaction. See “Who should not take Myalept?”
- benzyl alcohol toxicity. Serious side effects including death have happened in newborns or infants who have received the preservative benzyl alcohol. Myalept, when mixed with a liquid called bacteriostatic water for injection (BWFI), contains benzyl alcohol. Myalept, when mixed with a liquid called sterile water for injection (WFI) (preservative-free), contains no benzyl alcohol. When Myalept is used in newborns or infants, Myalept should be mixed with sterile water for injection (WFI).
The most common side effects of Myalept include:
- headache
- low blood sugar (hypoglycemia)
- decreased weight
- abdominal pain
Talk to your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the side effects with Myalept. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Myalept
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Myalept for a condition for which it was not prescribed. Do not give Myalept to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about Myalept. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Myalept that is written for health professionals.
For more information about Myalept, go to www.myalept.com or call Myalept Customer Service at 1-855-669-2537.
How should I store Myalept?
- Store Myalept in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep Myalept vials in their carton and out of the light.
- Do not freeze Myalept.
- Do not use Myalept past the expiration date printed on the vial.
- Do not use Myalept if the white powder in the vial is discolored.
- After mixing, the Myalept liquid in the vial should be clear and colorless. Do not use Myalept if it is colored or cloudy, or has any lumps or particles in it. Throw the vial away and get a new one.
- After mixing, do not freeze or shake Myalept.
- Myalept mixed with BWFI:
- Myalept can be used for more than 1 dose for up to 3 days when stored in the refrigerator between 36°F to 46°F (2°C to 8°C) and out of the light. Throw away any unused Myalept after 3 days.
- Myalept mixed with WFI:
- Should be used right away. Throw away any unused Myalept, it cannot be saved for later use.
Keep Myalept and all medicines out of the reach of children.
What are the ingredients in Myalept?
Active Ingredient: metreleptin
Inactive Ingredients: glutamic acid, glycine, sucrose, and polysorbate 20
Label
PRINCIPAL DISPLAY PANEL – 11.3 MG VIAL LABEL
- myalept®
(metreleptin) For Injection
11.3 mg per vial
Subcutaneous use only. - NDC 76431-210-01
Rx Only
Sterile - Single-dose vial if reconstituted with Water for Injection.
- Multi-dose vial if reconstituted with Bacteriostatic
Water for Injection. - Store refrigerated: 36°F to 46°F (2°C to 8°C).
Manufactured by: Amryt Pharmaceuticals DAC, Dublin, Ireland
US License No. 2208
PN 4084-02

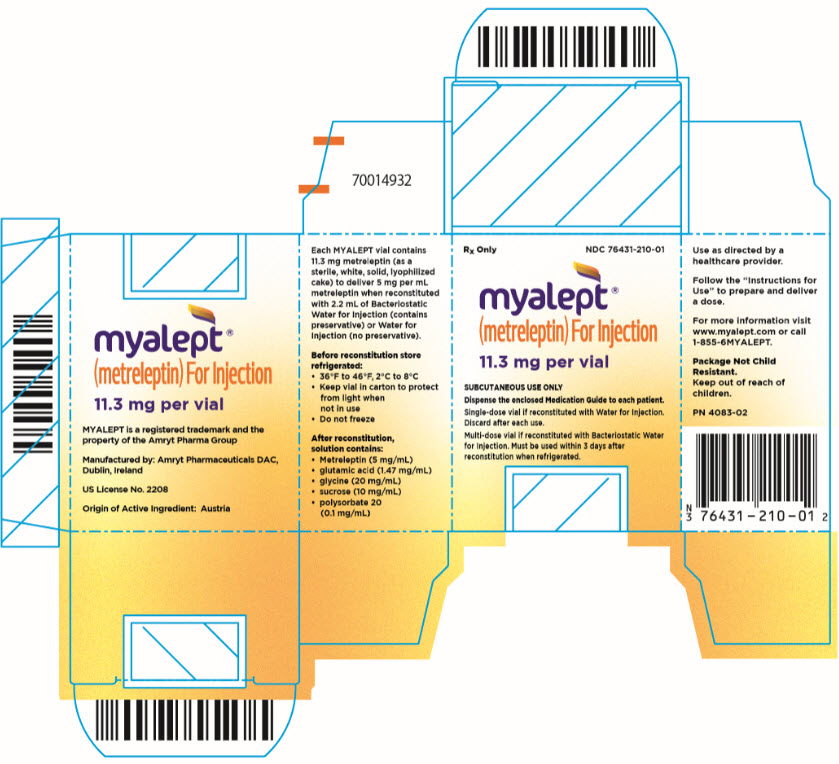
PRINCIPAL DISPLAY PANEL – 11.3 MG VIAL CARTON
- Rx Only
NDC 76431-210-01 - myalept®
(metreleptin) For Injection
11.3 mg per vial - SUBCUTANEOUS USE ONLY
- Dispense the enclosed Medication Guide to each patient.
- Single-dose vial if reconstituted with Water for Injection.
Discard after each use. - Multi-dose vial if reconstituted with Bacteriostatic Water
for Injection. Must be used within 3 days after
reconstitution when refrigerated.
SRC: NLM .
