Mavenclad
Generic name: cladribine (oral)
Drug class: Antimetabolites
Medically reviewed by A Ras MD.
What is Mavenclad?
Mavenclad is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Because of its safety profile, Mavenclad is generally used in people who have tried another MS medicine that they could not tolerate or that has not worked well enough.
Mavenclad is not recommended for use in people with clinically isolated syndrome (CIS).
It is not known if Mavenclad is safe and effective in children under 18 years of age.
Description
MAVENCLAD contains the nucleoside metabolic inhibitor cladribine, which is a white or almost white, non-hydroscopic, crystalline powder with the molecular formula C10H12ClN5O3 and molecular weight 285.69. It differs in structure from the naturally occurring nucleoside, deoxyadenosine, by the substitution of chlorine for hydrogen in the 2-position of the purine ring.
The chemical name of cladribine is 2-chloro-2′-deoxy-adenosine. The structural formula is shown below:
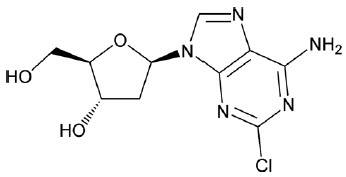
Cladribine is stable at slightly basic and at neutral pH. The main degradation pathway is hydrolysis and at acidic pH significant decomposition occurs with time. The ionization behavior of the molecule over the pH range 0 to 12 is characterized by a single pKa of approximately 1.21.
MAVENCLAD is provided as 10 mg tablets for oral use. Each MAVENCLAD 10 mg tablet contains cladribine as an active ingredient and hydroxypropyl betadex, magnesium stearate, and sorbitol as inactive ingredients.
Mechanism of Action
The mechanism by which cladribine exerts its therapeutic effects in patients with multiple sclerosis has not been fully elucidated but is thought to involve cytotoxic effects on B and T lymphocytes through impairment of DNA synthesis, resulting in depletion of lymphocytes.
What is the most important information I should know about Mavenclad?
Mavenclad can cause serious side effects, including:
- Risk of cancer (malignancies). Treatment with Mavenclad may increase your risk of developing cancer. Talk to your healthcare provider about your risk of developing cancer if you receive Mavenclad. You should follow your healthcare provider instructions about screening for cancer.
- Mavenclad may cause birth defects if used during pregnancy. Females must not be pregnant when they start treatment with Mavenclad or become pregnant during Mavenclad dosing and within 6 months after the last dose of each yearly treatment course. Stop your treatment with Mavenclad and call your healthcare provider right away if you become pregnant during treatment with Mavenclad.
- For females who are able to become pregnant:
- Your healthcare provider should order a pregnancy test for you before you begin your first and second yearly treatment course of Mavenclad to make sure that you are not pregnant. Your healthcare provider will decide when to do the test.
- Use effective birth control (contraception) on the days on which you take Mavenclad and for at least 6 months after the last dose of each yearly treatment course.
- Talk to your healthcare provider if you use oral contraceptives (the “pill”).
- You should use a second method of birth control on the days on which you take Mavenclad and for at least 4 weeks after your last dose of each yearly treatment course.
- For males with female partners who are able to become pregnant:
- Use effective birth control (contraception) during the days on which you take Mavenclad and for at least 6 months after the last dose of each yearly treatment course.
- For females who are able to become pregnant:
Who should not take Mavenclad?
Do not take Mavenclad if you:
- have cancer (malignancy).
- are pregnant, plan to become pregnant, or are a woman of childbearing age or a man able to father a child and you are not using birth control. See “What is the most important information I should know about Mavenclad?”
- are human immunodeficiency virus (HIV) positive.
- have active infections, including tuberculosis (TB), hepatitis B or C.
- are allergic to cladribine.
- are breastfeeding. See “What should I tell my healthcare provider before taking Mavenclad?”.
What should I tell my healthcare provider before taking Mavenclad?
Before you take Mavenclad, tell your healthcare provider about all of your medical conditions, including if you:
- think you have an infection.
- have heart failure.
- have liver or kidney problems.
- have taken, take, or plan to take medicines that affect your immune system or your blood cells, or other treatments for MS. Certain medicines can increase your risk of getting an infection.
- have had a recent vaccination or are scheduled to receive any vaccinations. You should not receive live or live-attenuated vaccines within the 4 to 6 weeks preceding your treatment with Mavenclad. You should not receive these types of vaccines during your treatment with Mavenclad and until your healthcare provider tells you that your immune system is no longer weakened.
- have or have had cancer.
- are breastfeeding or plan to breastfeed. It is not known if Mavenclad passes into your breast milk. Do not breastfeed on the days, on which you take Mavenclad, and for 10 days after the last dose. See “Who should not take Mavenclad?”.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I take Mavenclad?
- Mavenclad is given as two yearly treatment courses.
- Each yearly treatment course consists of 2 treatment weeks (also called cycles) that will be about a month apart. Your healthcare provider will tell you when you have to start your treatment weeks and how many tablets per week you need, depending on your weight. Each treatment week is 4 or 5 days.
- Your pharmacist will dispense a carton of Mavenclad for each treatment week. The prescribed number of tablets per day are provided in child resistant day packs.
- Take Mavenclad exactly as your healthcare provider tells you. Do not change your dose or stop taking Mavenclad unless your healthcare provider tells you to.
- Take Mavenclad with water and swallow whole without chewing. Mavenclad can be taken with or without food.
- Swallow Mavenclad right away after opening the blister pack.
- Your hands must be dry when handling Mavenclad and washed well with water afterwards.
- Limit contact with your skin. Avoid touching your nose, eyes and other parts of the body. If you get Mavenclad on your skin or on any surface, wash it right away with water.
- Take Mavenclad at least 3 hours apart from other medicines taken by mouth during the 4- to 5-day Mavenclad treatment week.
- If you miss a dose, take it as soon as you remember on the same day. If the whole day passes before you remember, take your missed dose the next day. Do not take 2 doses at the same time. Instead, you will extend the number of days in that treatment week.
Your healthcare provider will continue to monitor your health during the 2 yearly treatment courses, and for at least another 2 years during which you do not need to take Mavenclad. It is not known if Mavenclad is safe and effective in people who restart Mavenclad treatment more than 2 years after completing 2 yearly treatment courses.
What are the possible side effects of Mavenclad?
Mavenclad can cause serious side effects, including:
- See “What is the most important information I should know about Mavenclad?”
- low blood cell counts. Low blood cell counts have happened and can increase your risk of infections during your treatment with Mavenclad. Your healthcare provider will do blood tests before you start treatment with Mavenclad, during your treatment with Mavenclad, and afterward, as needed.
- serious infections such as:
- TB, hepatitis B or C, and shingles (herpes zoster). Fatal cases of TB and hepatitis have happened with cladribine during clinical studies. Tell your healthcare provider right away if you get any symptoms of the following infection related problems or if any of the symptoms get worse, including:
- fever
- aching painful muscles
- headache
- feeling of being generally unwell
- loss of appetite
- burning, tingling, numbness or itchiness of the skin in the affected area
- skin blotches, blistered rash and severe pain
- progressive multifocal leukoencephalopathy (PML). PML is a rare brain infection that usually leads to death or severe disability. Although PML has not been seen in MS patients taking Mavenclad, it may happen in people with weakened immune systems. Symptoms of PML get worse over days to weeks. Call your healthcare provider right away if you have any new or worsening neurologic signs or symptoms of PML, that have lasted several days, including:
- weakness on 1 side of your body
- loss of coordination in your arms and legs
- decreased strength
- problems with balance
- changes in your vision
- changes in your thinking or memory
- confusion
- changes in your personality
- TB, hepatitis B or C, and shingles (herpes zoster). Fatal cases of TB and hepatitis have happened with cladribine during clinical studies. Tell your healthcare provider right away if you get any symptoms of the following infection related problems or if any of the symptoms get worse, including:
- liver problems. Mavenclad may cause liver problems. Your healthcare provider should do blood tests to check your liver before you start taking Mavenclad. Call your healthcare provider right away if you have any of the following symptoms of liver problems:
- allergic reactions (hypersensitivities). Mavenclad can cause serious allergic reactions. Stop your treatment with Mavenclad and go to the closest emergency room for medical help right away if you have any signs or symptoms of allergic reactions. Symptoms of an allergic reaction may include: skin rash, swelling or itching of the face, lips, tongue or throat, or trouble breathing.
- heart failure. Mavenclad may cause heart failure, which means your heart may not pump as well as it should. Call your healthcare provider or go to the closest emergency room for medical help right away if you have any signs or symptoms such as shortness of breath, a fast or irregular heart beat, or unusual swelling in your body.
Your healthcare provider may delay or completely stop treatment with Mavenclad if you have severe side effects.
The most common side effects of Mavenclad include:
- upper respiratory infection
- headache
- low white blood cell counts
These are not all the possible side effects of Mavenclad. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Mavenclad
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Mavenclad for a condition for which it was not prescribed. Do not give Mavenclad to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider for information about Mavenclad that is written for health professionals.
How should I store Mavenclad?
- Mavenclad comes in a child resistant package.
- Store Mavenclad at room temperature between 68°F and 77°F (20°C and 25°C).
- Store Mavenclad in the original package to protect from moisture.
- Ask your healthcare provider or pharmacist about how to safely throw away any unused or expired Mavenclad tablets and packaging.
Keep Mavenclad and all medicines out of the reach of children.
What are the ingredients in Mavenclad?
Active ingredient: cladribine
Inactive ingredients: hydroxypropyl betadex, magnesium stearate, and sorbitol.
Label
PRINCIPAL DISPLAY PANEL – 4 TABLET BLISTER PACK CARTON
- NDC 44087-4000-4
- MAVENCLAD®
(cladribine) tablets
10 mg per tablet - Contents:
4 tablets: Four child-resistant day packs each
containing one 10 mg tablet in blister card. - Dosage and Administration:
Based on body weight. For oral administration.
See enclosed Prescribing Information and Medication
Guide. - Separate ingestion of other oral medicines by at
least 3 hours before and after taking MAVENCLAD. - Cytotoxic Agent:
Handle tablets as directed. - Dispense with enclosed Medication Guide.
- Rx Only
- 4 Tablets
EMD
SERONO
SRC: NLM .
