Invega Hafyera
Generic name: paliperidone palmitate
Dosage form: extended-release injectable suspension
Drug class: Atypical antipsychotics
What is Invega Hafyera?
Invega Hafyera is a prescription medicine given by injection by a healthcare provider 1 time every 6 months and used for the treatment of schizophrenia in adults who have been adequately treated with either:
- A 1 time each month paliperidone palmitate extended-release injectable suspension for at least 4 months.
- A 1 time every 3 months paliperidone palmitate extended-release injectable suspension for at least 3 months.
It is not known if Invega Hafyera is safe and effective in children under 18 years of age.
Description
Invega Hafyera contains a racemic mixture of (+)- and (-)- paliperidone palmitate. Paliperidone palmitate is an atypical antipsychotic belonging to the chemical class of benzisoxazole derivatives. The chemical name is (9RS)-3-[2-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)piperidin-1-yl]ethyl]-2-methyl-4-oxo-6,7,8,9-tetrahydro-4H-pyrido[1,2-a]pyrimadin-9-yl hexadecanoate. Its molecular formula is C39H57FN4O4 and its molecular weight is 664.89. The structural formula is:
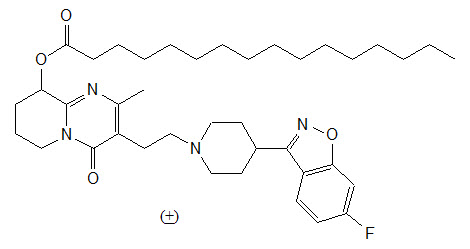
Paliperidone palmitate is very slightly soluble in ethanol and methanol, practically insoluble in polyethylene glycol 400 and propylene glycol, and slightly soluble in ethyl acetate.
Mechanism of Action
Paliperidone palmitate is hydrolyzed to paliperidone. Paliperidone is the major active metabolite of risperidone. The mechanism of action of paliperidone is unclear. However, its efficacy in the treatment of schizophrenia could be mediated through a combination of central dopamine D2 and serotonin 5HT2A receptor antagonism.
What are the ingredients in Invega Hafyera?
Active ingredient: paliperidone palmitate
Inactive ingredients: polysorbate 20, polyethylene glycol 4000, citric acid monohydrate, sodium dihydrogen phosphate monohydrate, sodium hydroxide, and water for injection
What is the most important information I should know about Invega Hafyera?
Invega Hafyera may cause serious side effects, including:
- Increased risk of death in elderly people with dementia-related psychosis. Invega Hafyera increases the risk of death in elderly people who have lost touch with reality (psychosis) due to confusion and memory loss (dementia). Invega Hafyera is not for the treatment of people with dementia-related psychosis.
Who should not use Invega Hafyera?
Do not receive Invega Hafyera if you are allergic to paliperidone palmitate, risperidone, or any of the ingredients in Invega Hafyera. See the end of this Patient Information leaflet for a complete list of ingredients in Invega Hafyera.
What should I tell my healthcare provider before using Invega Hafyera
Before receiving Invega Hafyera, tell your healthcare provider about all your medical conditions, including if you:
- have had Neuroleptic Malignant Syndrome (NMS)
- have or have had heart problems, including a heart attack, heart failure, abnormal heart rhythm, or long QT syndrome
- have or have had low levels of potassium or magnesium in your blood
- have or have had uncontrolled movements of your tongue, face, mouth, or jaw (tardive dyskinesia)
- have or have had kidney or liver problems
- have diabetes or have a family history of diabetes
- have Parkinson’s disease or a type of dementia called Lewy Body Dementia
- have had a low white blood cell count
- have had problems with dizziness or fainting or are being treated for high blood pressure
- have or have had seizures or epilepsy
- are pregnant or plan to become pregnant. It is not known if Invega Hafyera will harm your unborn baby.
- Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with Invega Hafyera.
- If you become pregnant while receiving Invega Hafyera, talk to your healthcare provider about registering with the National Pregnancy Registry for Atypical Antipsychotics. You can register by calling 1-866-961-2388 or visit http://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/.
- Babies born to mothers who receive Invega Hafyera during their third trimester of pregnancy may develop agitation, low muscle tone (floppy baby syndrome) tremors, excessive sleepiness, breathing problems, and feeding problems. Tell your healthcare provider right away if your baby develops any of these symptoms.
- are breastfeeding or plan to breastfeed. Invega Hafyera can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with Invega Hafyera.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Invega Hafyera and other medicines may affect each other causing possible serious side effects. Invega Hafyera may affect the way other medicines work, and other medicines may affect how Invega Hafyera works.
Your healthcare provider can tell you if it is safe to receive Invega Hafyera with your other medicines. Do not start or stop any medicines during treatment with Invega Hafyera without talking to your healthcare provider first.
Know the medicines you take. Keep a list of them to show to your healthcare provider or pharmacist when you get a new medicine.
How should I use Invega Hafyera?
- Follow your Invega Hafyera treatment schedule exactly as your healthcare provider tells you to.
- Your healthcare provider will tell you how much Invega Hafyera you will receive and when you will receive it.
- Invega Hafyera is given as an injection by your healthcare provider into the muscle (intramuscularly) of your buttocks, 1 time every 6 months.
What should I avoid while using Invega Hafyera?
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how Invega Hafyera affects you. Invega Hafyera may affect your judgment, thinking, or motor skills.
- Avoid getting too hot or dehydrated.
- Do not exercise too much.
- In hot weather, stay inside in a cool place if possible.
- Stay out of the sun.
- Do not wear too much clothing or heavy clothing.
- Drink plenty of water.
What are the possible side effects of Invega Hafyera?
Invega Hafyera may cause serious side effects, including:
- Cerebrovascular problems (including stroke) in elderly people with dementia-related psychosis that can lead to death.
- Neuroleptic Malignant Syndrome (NMS), a serious condition that can lead to death. Call your healthcare provider or go to your nearest hospital emergency room right away if you have some or all of the following signs and symptoms of NMS:
- high fever
- confusion
- changes in your breathing, pulse, heart rate, and blood pressure
- stiff muscles
- sweating
- Problems with your heartbeat. These heart problems can cause death. Call your healthcare provider right away if you have any of these symptoms:
- passing out or feeling like you will pass out
- dizziness
- feeling as if your heart is pounding or missing beats
- Uncontrolled body movements (tardive dyskinesia). Invega Hafyera may cause movements that you cannot control in your face, tongue, or other body parts. Tardive dyskinesia may not go away, even if you stop receiving Invega Hafyera. Tardive dyskinesia may also start after you stop receiving Invega Hafyera.
- Problems with your metabolism such as:
- high blood sugar (hyperglycemia) and diabetes. Increases in blood sugar can happen in some people who receive Invega Hafyera. Extremely high blood sugar can lead to coma or death. Your healthcare provider should check your blood sugar before you start and regularly during treatment with Invega Hafyera.
Call your healthcare provider if you have any of these symptoms of high blood sugar during treatment with Invega Hafyera:- feel very thirsty
- feel very hungry
- feel sick to your stomach
- need to urinate more than usual
- feel weak or tired
- feel confused, or your breath smells fruity
- increased fat levels (cholesterol and triglycerides) in your blood. Your healthcare provider should check the fat levels in your blood before you start and regularly during treatment with Invega Hafyera.
- weight gain. You and your healthcare provider should check your weight before you start and often during treatment with Invega Hafyera.
- high blood sugar (hyperglycemia) and diabetes. Increases in blood sugar can happen in some people who receive Invega Hafyera. Extremely high blood sugar can lead to coma or death. Your healthcare provider should check your blood sugar before you start and regularly during treatment with Invega Hafyera.
- Decreased blood pressure (orthostatic hypotension) and fainting. You may feel lightheaded or faint when you rise too quickly from a sitting or lying position, especially early in treatment or when the dose is changed.
- Falls. Invega Hafyera may make you sleepy or dizzy, may cause a decrease in your blood pressure when changing position (orthostatic hypotension), and can slow your thinking and motor skills which may lead to falls that can cause fractures or other injuries.
- Low white blood cell count. Your healthcare provider may do blood tests during the first few months of treatment with Invega Hafyera.
- Increased prolactin levels in your blood (hyperprolactinemia). Invega Hafyera may cause a rise in the blood levels of a hormone called prolactin (hyperprolactinemia) that may cause side effects including missed menstrual periods, a reversible reduction in fertility in females who are able to become pregnant, leakage of milk from the breasts, development of breasts in men, or problems with erection.
- Invega Hafyera can make you sleepy or dizzy, and can slow your thinking and motor skills. Do not drive, operate heavy machinery, or do other dangerous activities until you know how Invega Hafyera affects you.
- Seizures (convulsions).
- Difficulty swallowing that can cause food or liquid to get into your lungs.
- Prolonged or painful erection lasting more than 4 hours (priapism). Call your healthcare provider or go to your nearest emergency room right away if you have an erection that lasts more than 4 hours.
The most common side effects of Invega Hafyera include:
- upper respiratory tract infections
- weight gain
- feeling restlessness or difficulty sitting still
- tremors
- shuffling walk
- injection site reactions
- headache
- slow movements
- stiffness
These are not all the possible side effects of Invega Hafyera.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How supplied/stored
INVEGA HAFYERA™ is available as a white to off-white sterile aqueous extended-release suspension for gluteal intramuscular injection in dose strengths of 1,092 mg/3.5 mL and 1,560 mg/5 mL paliperidone palmitate. The kit contains a single-dose prefilled syringe and a 20G, 1½-inch safety needle.
1,092 mg paliperidone palmitate kit (NDC 50458-611-01)
1,560 mg paliperidone palmitate kit (NDC 50458-612-01)
Storage and Handling
Store at room temperature 20°C to 25°C (68°F to 77°F); excursions between 15°C and 30°C (59°F and 86°F) are permitted. Do not mix with any other product or diluent.
Ship and store in a horizontal position. See arrows on product carton for proper orientation
Label
PRINCIPAL DISPLAY PANEL – 3.5 ML SYRINGE CARTON
- NDC 50458-611-01
- Single-dose prefilled syringe. Use entire contents of syringe.
- INVEGA HAFYERA™
(paliperidone palmitate)
extended-release injectable suspension - FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
- Each injection must be administered only by
a healthcare professional. - Shake before using.
- CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle). - Recommended Dosage: See Prescribing Information.
- Rx only
- Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted. - SHIP AND STORE
THIS SIDE UP LAY FLAT - 1,092 mg/3.5 mL
- Each single-dose prefilled syringe contains
1,092 mg paliperidone palmitate. - 6 MONTHS Administer
every 6 months - Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds

PRINCIPAL DISPLAY PANEL – 5 ML SYRINGE CARTON
- NDC 50458-612-01
- Single-dose prefilled syringe. Use entire contents of syringe.
- INVEGA HAFYERA™ (paliperidone palmitate) extended-release injectable suspension
- FOR GLUTEAL INTRAMUSCULAR INJECTION ONLY
- Each injection must be administered only by
a healthcare professional. - Shake before using.
- CONTENTS: 1 single-dose prefilled syringe and
1 needle (a 20G,1.5-inch thin wall safety needle). - Recommended Dosage: See Prescribing Information.
- Rx only
- Store at room temperature 20°C to 25°C
(68°F to 77°F); excursions between
15°C and 30°C (59°F and 86°F) are permitted. - SHIP AND STORE
THIS SIDE UP LAY FLAT - 1,560 mg/5 mL
- Each single-dose prefilled syringe contains
1,560 mg paliperidone palmitate. - 6 MONTHS Administer every 6 months
- Shake syringe
with the syringe
tip cap pointing
up VERY FAST
for at least
15 seconds, rest
briefly, then
shake again for
15 seconds

SRC: NLM .
