Breo Ellipta
Generic name: fluticasone and vilanterol
Drug class: Bronchodilator combinations
Medically reviewed by A Ras MD.
What is Breo Ellipta?
Breo Ellipta combines an inhaled corticosteroid (ICS) medicine, fluticasone furoate, and a long-acting beta2-adrenergic agonist (LABA) medicine, vilanterol, ICS medicines such as fluticasone furoate help to decrease inflammation in the lungs. Inflammation in the lungs can lead to breathing problems, LABA medicines such as vilanterol help the muscles around the airways in your lungs stay relaxed to prevent symptoms, such as wheezing, cough, chest tightness, and shortness of breath. These symptoms can happen when the muscles around the airways tighten. This makes it hard to breathe.
Breo Ellipta is not used to relieve sudden breathing problems and will not replace a rescue inhaler. Breo Ellipta should not be used in children and adolescents. It is not known if Breo Ellipta is safe and effective in children and adolescents younger than 18 years of age. Breo Ellipta is used for COPD and asthma and as follows:
- COPD: Breo Ellipta 100/25 is a prescription medicine used to treat COPD. COPD is a chronic lung disease that includes chronic bronchitis, emphysema, or both. Breo Ellipta 100/25 is used long term as 1 inhalation 1 time each day to improve symptoms of COPD for better breathing and to reduce the number of flare-ups (the worsening of your COPD symptoms for several days).
- Asthma: Breo Ellipta is a prescription medicine used as 1 inhalation 1 time each day to prevent and control symptoms of asthma for better breathing and to prevent symptoms such as wheezing.
- Breo Ellipta contains vilanterol. LABA medicines such as vilanterol when used alone increase the risk of hospitalizations and death from asthma problems. Breo Ellipta contains an ICS and a LABA. When an ICS and LABA are used together, there is not a significant increased risk in hospitalizations and death from asthma problems.
- Breo Ellipta is not for people with asthma who are well controlled with an asthma control medicine, such as a low to medium dose of an ICS medicine. Breo Ellipta is for adults with asthma who need both an ICS and LABA medicine.
Description
BREO ELLIPTA 100/25 and BREO ELLIPTA 200/25 are inhalation powders for oral inhalation that contain a combination of fluticasone furoate (an ICS) and vilanterol (a LABA).
One active component of BREO ELLIPTA is fluticasone furoate, a synthetic trifluorinated corticosteroid having the chemical name (6α,11β,16α,17α)-6,9-difluoro-17-{[(fluoro-methyl)thio]carbonyl}-11-hydroxy-16-methyl-3-oxoandrosta-1,4-dien-17-yl 2-furancarboxylate and the following chemical structure:
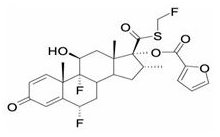
Fluticasone furoate is a white powder with a molecular weight of 538.6, and the empirical formula is C27H29F3O6S. It is practically insoluble in water.
The other active component of BREO ELLIPTA is vilanterol trifenatate, a LABA with the chemical name triphenylacetic acid-4-{(1R)-2-[(6-{2-[2,6-dicholorobenzyl)oxy]ethoxy}hexyl)amino]-1-hydroxyethyl}-2-(hydroxymethyl)phenol (1:1) and the following chemical structure:
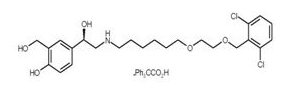
Vilanterol trifenatate is a white powder with a molecular weight of 774.8, and the empirical formula is C24H33Cl2NO5•C20H16O2. It is practically insoluble in water.
BREO ELLIPTA is a light grey and pale blue plastic inhaler containing 2 foil blister strips. Each blister on one strip contains a white powder mix of micronized fluticasone furoate (100 or 200 mcg) and lactose monohydrate (12.4 or 12.3 mg), and each blister on the other strip contains a white powder mix of micronized vilanterol trifenatate (40 mcg equivalent to 25 mcg of vilanterol), magnesium stearate (125 mcg), and lactose monohydrate (12.34 mg). The lactose monohydrate contains milk proteins. After the inhaler is activated, the powder within both blisters is exposed and ready for dispersion into the airstream created by the patient inhaling through the mouthpiece.
Under standardized in vitro test conditions, BREO ELLIPTA delivers 92 and 184 mcg of fluticasone furoate and 22 mcg of vilanterol per blister when tested at a flow rate of 60 L/min for 4 seconds.
In adult subjects with obstructive lung disease and severely compromised lung function (COPD with FEV1/FVC <70% and FEV1 <30% predicted or FEV1 <50% predicted plus chronic respiratory failure), mean peak inspiratory flow through the ELLIPTA inhaler was 66.5 L/min (range: 43.5 to 81.0 L/min).
In adult subjects with severe asthma, mean peak inspiratory flow through the ELLIPTA inhaler was 96.6 L/min (range: 72.4 to 124.6 L/min).
The actual amount of drug delivered to the lung will depend on patient factors, such as inspiratory flow profile.
Mechanism of Action
BREO ELLIPTA
Since BREO ELLIPTA contains both fluticasone furoate and vilanterol, the mechanisms of action described below for the individual components apply to BREO ELLIPTA. These drugs represent 2 different classes of medications (a synthetic corticosteroid and a LABA) that have different effects on clinical and physiological indices.
Fluticasone Furoate
Fluticasone furoate is a synthetic trifluorinated corticosteroid with anti-inflammatory activity. Fluticasone furoate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor that is approximately 29.9 times that of dexamethasone and 1.7 times that of fluticasone propionate. The clinical relevance of these findings is unknown.
The precise mechanism through which fluticasone furoate affects COPD and asthma symptoms is not known. Inflammation is an important component in the pathogenesis of COPD and asthma. Corticosteroids have been shown to have a wide range of actions on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines) involved in inflammation. Specific effects of fluticasone furoate demonstrated in in vitro and in vivo models included activation of the glucocorticoid response element, inhibition of pro-inflammatory transcription factors such as NFkB, and inhibition of antigen-induced lung eosinophilia in sensitized rats. These anti-inflammatory actions of corticosteroids may contribute to their efficacy.
Vilanterol
Vilanterol is a LABA. In vitro tests have shown the functional selectivity of vilanterol was similar to salmeterol. The clinical relevance of this in vitro finding is unknown.
Although beta2-receptors are the predominant adrenergic receptors in bronchial smooth muscle and beta1-receptors are the predominant receptors in the heart, there are also beta2-receptors in the human heart comprising 10% to 50% of the total beta-adrenergic receptors. The precise function of these receptors has not been established, but they raise the possibility that even highly selective beta2-agonists may have cardiac effects.
The pharmacologic effects of beta2-adrenoceptor agonist drugs, including vilanterol, are at least in part attributable to stimulation of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3′,5′-adenosine monophosphate (cyclic AMP). Increased cyclic AMP levels cause relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
Who should not take Breo Ellipta?
- to treat sudden, severe symptoms of asthma or COPD.
- if you have a severe allergy to milk proteins. Ask your healthcare provider if you are not sure.
- if you are allergic to fluticasone furoate, vilanterol, or any of the ingredients in Breo Ellipta. See the end of this patient information for a complete list of ingredients in Breo Ellipta.
What should I tell my healthcare provider before taking Breo Ellipta?
Before using Breo Ellipta, tell your healthcare provider about all of your medical conditions, including if you:
- have heart problems.
- have high blood pressure.
- have seizures.
- have thyroid problems.
- have diabetes or have been told you have high blood sugar.
- have liver problems.
- have weak bones (osteoporosis).
- have an immune system problem.
- have eye problems such as glaucoma, increased pressure in your eye, cataracts, or other changes in vision.
- are allergic to milk proteins.
- have any type of viral, bacterial, fungal, or parasitic infection.
- are exposed to chickenpox or measles.
- are pregnant or plan to become pregnant. It is not known if Breo Ellipta may harm your unborn baby.
- are breastfeeding. It is not known if the medicines in Breo Ellipta pass into your breast milk and if they can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Breo Ellipta and certain other medicines may interact with each other. This may cause serious side effects. Especially tell your healthcare provider if you take antifungal or anti-HIV medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Breo Ellipta?
Read the step-by-step instructions for using Breo Ellipta that come with your medication.
- Do not use Breo Ellipta unless your healthcare provider has taught you how to use the inhaler and you understand how to use it correctly.
- Breo Ellipta comes in 2 different strengths. Your healthcare provider prescribed the strength that is best for you.
- Use Breo Ellipta exactly as your healthcare provider tells you to use it. Do not use Breo Ellipta more often than prescribed.
- Use 1 inhalation of Breo Ellipta 1 time each day. Use Breo Ellipta at the same time each day.
- If you miss a dose of Breo Ellipta, take it as soon as you remember. Do not take more than 1 inhalation per day. Take your next dose at your usual time. Do not take 2 doses at 1 time.
- If you take too much Breo Ellipta, call your healthcare provider or go to the nearest hospital emergency room right away if you have any unusual symptoms, such as worsening shortness of breath, chest pain, increased heart rate, or shakiness.
- Do not use other medicines that contain a LABA for any reason. Ask your healthcare provider or pharmacist if any of your other medicines are LABA medicines.
- Do not stop using Breo Ellipta unless told to do so by your healthcare provider because your symptoms might get worse. Your healthcare provider will change your medicines as needed.
- Breo Ellipta does not relieve sudden symptoms of asthma or COPD and you should not take extra doses of Breo Ellipta to relieve these sudden symptoms. Always have a rescue inhaler with you to treat sudden symptoms. If you do not have a rescue inhaler, call your healthcare provider to have one prescribed for you.
- Call your healthcare provider or get medical care right away if:
- your breathing problems get worse.
- you need to use your rescue inhaler more often than usual.
- your rescue inhaler does not work as well to relieve your symptoms.
- your peak flow meter results decrease. Your healthcare provider will tell you the numbers that are right for you.
What are the possible side effects of Breo Ellipta?
Breo Ellipta can cause serious side effects, including:
- fungal infection in your mouth or throat (thrush). Rinse your mouth with water without swallowing after using Breo Ellipta to help reduce your chance of getting thrush.
- pneumonia. People with COPD have a higher chance of getting pneumonia. Breo Ellipta may increase the chance of getting pneumonia. Call your healthcare provider if you notice any of the following symptoms:
- increase in mucus (sputum) production
- change in mucus color
- fever
- chills
- increased cough
- increased breathing problems
- weakened immune system and increased chance of getting infections (immunosuppression).
- reduced adrenal function (adrenal insufficiency). Adrenal insufficiency is a condition where the adrenal glands do not make enough steroid hormones. This can happen when you stop taking oral corticosteroid medicines (such as prednisone) and start taking a medicine containing an ICS (such as Breo Ellipta). During this transition period, when your body is under stress from fever, trauma (such as a car accident), infection, surgery, or worse COPD symptoms, adrenal insufficiency can get worse and may cause death. Symptoms of adrenal insufficiency include:
- sudden breathing problems immediately after inhaling your medicine. If you have sudden breathing problems immediately after inhaling your medicine, stop taking Breo Ellipta and call your healthcare provider right away.
- serious allergic reactions. Call your healthcare provider or get emergency medical care if you get any of the following symptoms of a serious allergic reaction:
- rash
- hives
- swelling of your face, mouth, and tongue
- breathing problems
- effects on heart:
- increased blood pressure
- a fast or irregular heartbeat, awareness of heartbeat
- chest pain
- effects on nervous system:
- tremor
- nervousness
- bone thinning or weakness (osteoporosis).
- eye problems including glaucoma, increased pressure in your eye, cataracts, or other changes in vision. You should have regular eye exams while using Breo Ellipta.
- high levels of blood sugar (hyperglycemia), which may cause increased thirst, frequent urination, or unexplained tiredness.
- changes in laboratory blood levels, including low levels of potassium (hypokalemia).
- slowed growth in children.
Common side effects of Breo Ellipta include:
COPD:
- runny nose and sore throat
- upper respiratory tract infection
- headache
- thrush in your mouth or throat. Rinse your mouth with water without swallowing after use to help prevent this.
- back pain
- pneumonia
- bronchitis
- inflammation of the sinuses
- cough
- mouth and throat pain
- joint pain
- increased blood pressure
- flu
- fever
Asthma:
- runny nose and sore throat
- thrush in your mouth or throat. Rinse your mouth with water without swallowing after use to help prevent this.
- headache
- flu
- respiratory tract infection
- bronchitis
- inflammation of the sinuses
- mouth and throat pain
- hoarseness and voice changes
- cough
These are not all the possible side effects of Breo Ellipta.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Breo Ellipta
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Breo Ellipta for a condition for which it was not prescribed. Do not give Breo Ellipta to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about Breo Ellipta that is written for health professionals.
How should I store Breo Ellipta?
- Store Breo Ellipta at room temperature between 68°F and 77°F (20°C and 25°C). Keep in a dry place away from heat and sunlight.
- Store Breo Ellipta in the unopened tray and only open when ready for use.
- Safely throw away Breo Ellipta in the trash 6 weeks after you open the tray or when the counter reads “0”, whichever comes first. Write the date you open the tray on the label on the inhaler.
Keep Breo Ellipta and all medicines out of the reach of children.
What are the ingredients in Breo Ellipta?
Active ingredients: fluticasone furoate, vilanterol trifenatate
Inactive ingredients: lactose monohydrate (contains milk proteins), magnesium stearate
Label
PRINCIPAL DISPLAY PANEL
- NDC 0173-0882-10
- BREO ELLIPTA 200/25
- (fluticasone furoate 200 mcg and vilanterol 25 mcg inhalation powder)
- 200 mcg/25 mcg
- Rx Only
- FOR ORAL INHALATION ONLY
- BREO ELLIPTA contains 2 foil strips of 30 blisters each. Each blister on one strip contains 200 mcg of fluticasone furoate and lactose monohydrate. Each blister on the other strip contains 25 mcg of vilanterol, magnesium stearate, and lactose monohydrate.
- 1 ELLIPTA Inhaler containing 30 doses (60 blisters total)
- Trademarks are owned by or licensed to the GSK group of Companies. BREO ELLIPTA was developed in collaboration with Innoviva, Inc.
- ©2022 GSK group of companies or its licensor.
- 62000000074680 Rev. 1/22
-

SRC: NLM .
