Verquvo
Generic name: vericiguat
Drug class: Vasodilators
Medically reviewed by A Ras MD.
What is Verquvo?
Verquvo is a prescription medicine used in adults who are having symptoms of their chronic (long-lasting) heart failure, who have had a recent hospitalization or the need to receive intravenous (IV) medicines and have an ejection fraction (amount of blood pumped with each heartbeat) of less than 45 percent to reduce the risk of dying and to reduce the need to be hospitalized
Heart failure happens when your heart is weak and cannot pump enough blood to your lungs and the rest of your body.
It is not known if Verquvo is safe and effective in children.
Description
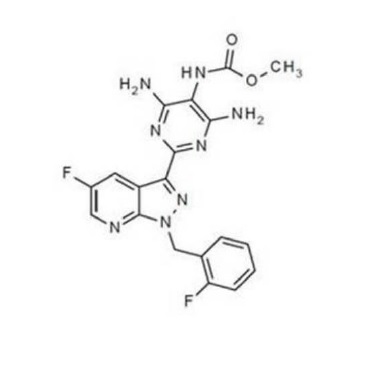
VERQUVO tablets contains vericiguat, a soluble guanylate cyclase stimulator.
The chemical name of vericiguat is methyl {4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1H-pyrazolo[3,4-b] pyridin-3-yl] pyrimidin-5-yl} carbamate. The molecular formula is C19H16F2N8O2 and the molecular weight is 426.39 g/mol.
The chemical structure is:
 |
Vericiguat is a white to yellowish powder that is freely soluble in dimethyl sulfoxide; slightly soluble in acetone; very slightly soluble in ethanol, acetonitrile, methanol, and ethyl acetate; and practically insoluble in 2-propanol.
VERQUVO® is available as film-coated tablets for oral administration, containing 2.5 mg of vericiguat, 5 mg of vericiguat or 10 mg of vericiguat.
The tablet inactive ingredients are croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate.
The film coating contains hypromellose, talc and titanium dioxide. The film coating for the 5 mg of VERQUVO tablet also contains ferric oxide red. The film coating for the 10 mg of VERQUVO tablet also contains ferric oxide yellow.
Mechanism of Action
Vericiguat is a stimulator of soluble guanylate cyclase (sGC), an important enzyme in the nitric oxide (NO) signaling pathway. When NO binds to sGC, the enzyme catalyzes the synthesis of intracellular cyclic guanosine monophosphate (cGMP), a second messenger that plays a role in the regulation of vascular tone, cardiac contractility, and cardiac remodeling. Heart failure is associated with impaired synthesis of NO and decreased activity of sGC, which may contribute to myocardial and vascular dysfunction. By directly stimulating sGC, independently of and synergistically with NO, vericiguat augments levels of intracellular cGMP, leading to smooth muscle relaxation and vasodilation.
What is the most important information I should know about Verquvo?
Verquvo may cause birth defects if taken during pregnancy.
- Females must not be pregnant when they start taking Verquvo.
- Females who are able to get pregnant:
- Your healthcare provider will do a pregnancy test to make sure that you are not pregnant before you start taking Verquvo.
- You must use effective forms of birth control during treatment and for 1 month after you stop treatment with Verquvo. Talk to your healthcare provider about forms of birth control that you may use to prevent pregnancy during treatment with Verquvo.
- Tell your healthcare provider right away if you become pregnant or think you are pregnant during treatment with Verquvo.
Who should not take Verquvo?
Do not take Verquvo if you:
- are taking another medicine called a soluble guanylate cyclase stimulator (sGC). Ask your healthcare provider if you are not sure if you are taking an sGC medicine.
- are pregnant. See“What is the most important information I should know about Verquvo?”
What should I tell my healthcare provider before taking Verquvo?
Before you take Verquvo, tell your healthcare provider about all your medical conditions, including if you:
- are breastfeeding or plan to breastfeed. It is not known if Verquvo passes into your breast milk. Do not breastfeed if you take Verquvo. Talk with your healthcare provider about the best way to feed your baby if you take Verquvo.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Certain other medicines may affect how Verquvo works.
How should I take Verquvo?
- Take Verquvo exactly as your healthcare provider tells you to.
- Take Verquvo 1 time each day with food.
- Swallow Verquvo tablets whole. If you are not able to swallow the tablet whole, you may crush Verquvo tablets and mix with water right before taking your dose.
- Your healthcare provider may change your dose — when you first start taking Verquvo to find the best dose for you and how well you tolerate Verquvo.
- If you miss a dose, take the missed dose as soon as you remember on the same day of the missed dose. Do not take 2 doses of Verquvo on the same day to make up for a missed dose.
- If you take too much Verquvo, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Verquvo?
Verquvo may cause serious side effects, including:
See“What is the most important information I should know about Verquvo?”
The most common side effects of Verquvo include:
- low blood pressure
- low red blood cells (anemia)
These are not all the possible side effects of Verquvo. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Verquvo
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Verquvo for a condition for which it was not prescribed. Do not give Verquvo to other people, even if they have the same symptoms you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about Verquvo that is written for health professionals.
How should I store Verquvo?
- Store Verquvo at room temperature between 68°F to 77°F (20°C to 25°C).
Keep Verquvo and all medicines out of the reach of children.
What are the ingredients in Verquvo?
Active ingredient: vericiguat.
Inactive ingredients: croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate.
The tablet film coating contains: hypromellose, talc, titanium dioxide. The film-coating for the 5 mg tablet also contains ferric oxide red. The film-coating for the 10 mg tablet also contains ferric oxide yellow.
Label
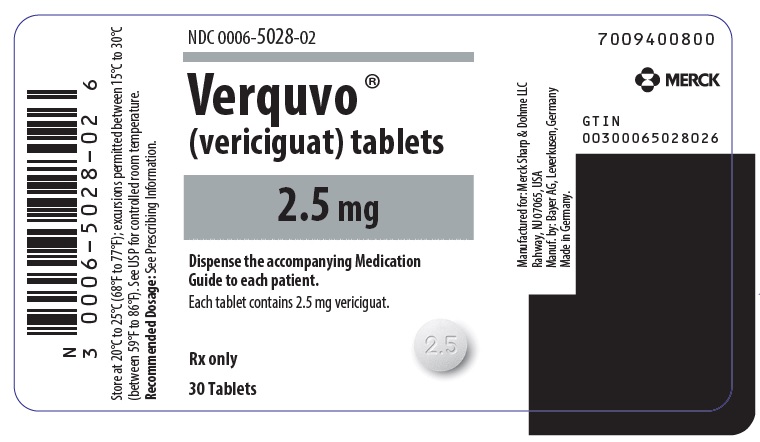
PRINCIPAL DISPLAY PANEL – 2.5 MG TABLET BOTTLE LABEL
- NDC 0006-5028-02
- Verquvo ®
- (vericiguat) tablets
- 2.5 mg
- Dispense the accompanying Medication
Guide to each patient. - Each tablet contains 2.5 mg vericiguat.
- Rx only
- 30 Tablets
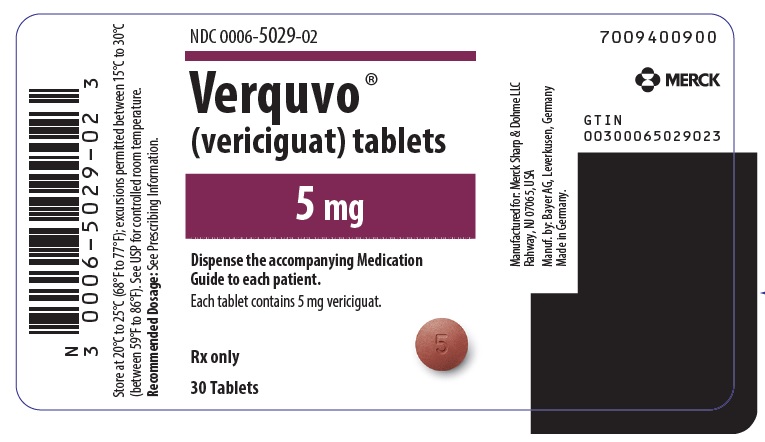
PRINCIPAL DISPLAY PANEL – 5 MG TABLET BOTTLE LABEL
- NDC 0006-5029-02
- Verquvo ®
- (vericiguat) tablets
- 5 mg
- Dispense the accompanying Medication
Guide to each patient. - Each tablet contains 5 mg vericiguat.
- Rx only
- 30 Tablets
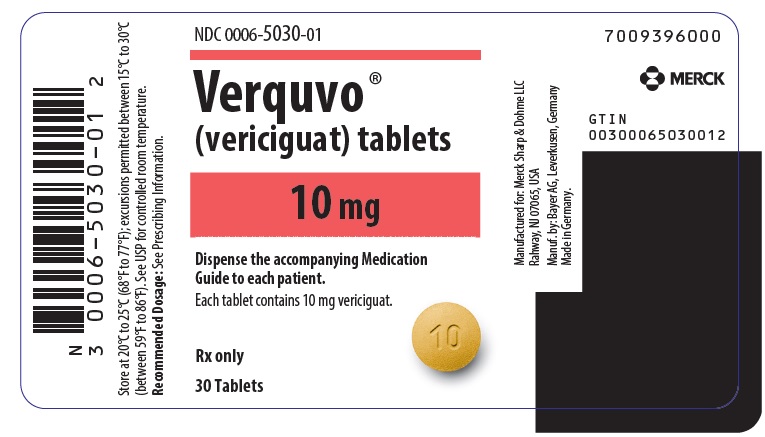
PRINCIPAL DISPLAY PANEL – 10 MG TABLET BOTTLE LABEL
- NDC 0006-5030-01
- Verquvo ®
- (vericiguat) tablets
- 10 mg
- Dispense the accompanying Medication
Guide to each patient. - Each tablet contains 10 mg vericiguat.
- Rx only
- 30 Tablets
SRC: NLM .
