Rukobia
Generic name: fostemsavir (extended-release tablets)
Drug class: Miscellaneous antivirals
Medically reviewed by A Ras MD.
What is Rukobia?
Rukobia is a prescription medicine that is used with other antiretroviral medicines to treat Human Immunodeficiency Virus (HIV-1) infection in adults who have received several anti-HIV-1 regimens in the past, and have HIV-1 virus that is resistant to many antiretroviral medicines, and are failing their current antiretroviral therapy. You could be failing therapy because it is not working or no longer works, you are not able to tolerate the side effects, or there are other safety reasons why you cannot take it.
HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS).
It is not known if Rukobia is safe and effective in children.
Description
Fostemsavir tromethamine is a prodrug of temsavir, an HIV-1 gp120-directed attachment inhibitor.
The chemical name of fostemsavir tromethamine is (3-((4-benzoyl-1-piperazinyl)(oxo)acetyl)-4-methoxy-7-(3-methyl-1H-1,2,4-triazol-1-yl)-1H-pyrrolo[2,3-c]pyridin-1-yl)methyl dihydrogen phosphate, 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1). The empirical formula is C25H26N7O8P•C4H11NO3. The molecular weight is 704.6 g/mol (583.5 as free acid). It has the following structural formula:
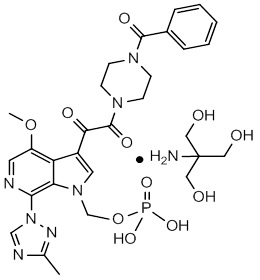
Fostemsavir tromethamine is a white powder and is soluble to greater than 250 mg/mL in aqueous solutions with a pH greater than 3.7.
RUKOBIA extended-release tablets are for oral administration. Each film-coated tablet contains 600 mg of fostemsavir (equivalent to 725 mg fostemsavir tromethamine), and the following inactive ingredients: colloidal silicon dioxide, hydroxypropyl cellulose, hypromellose, and magnesium stearate. The tablet film-coating contains the inactive ingredients iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
Who should not take Rukobia?
Do not take Rukobia if you:
- are allergic to fostemsavir or any of the ingredients in Rukobia. See the end of this guide for a complete list of ingredients in Rukobia.
- take certain medicines, including:
- enzalutamide
- carbamazepine
- phenytoin
- rifampin
- mitotane
- St. John’s wort (Hypericum perforatum)
What should I tell my healthcare provider before taking Rukobia?
Before taking Rukobia, tell your healthcare provider about all of your medical conditions, including if you:
- have or have had a heart problem, including a heart rhythm problem called QTc prolongation (irregular heartbeat).
- have or have had liver problems, including hepatitis B or C virus infection.
- are pregnant or plan to become pregnant. It is not known if Rukobia will harm your unborn baby. Tell your healthcare provider if you become pregnant during treatment with Rukobia.
Pregnancy Exposure Registry. There is a pregnancy exposure registry for women who take antiretroviral medicines, including Rukobia, during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk to your healthcare provider about how you can take part in this registry. - are breastfeeding or plan to breastfeed. Do not breastfeed if you take Rukobia.
- You should not breastfeed if you have HIV-1 because of the risk of passing HIV-1 to your baby.
- It is not known if Rukobia can pass to your baby in your breast milk.
Talk with your healthcare provider about the best way to feed your baby during treatment with Rukobia.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Some medicines interact with Rukobia.
Especially tell your healthcare provider if you take birth control pills (oral contraceptives) that contain ethinyl estradiol. The amount of ethinyl estradiol can become increased in your blood during treatment with Rukobia. Talk to your healthcare provider about which oral contraceptives may be right for you during treatment with Rukobia.
Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with Rukobia.
- Do not start taking a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take Rukobia with other medicines.
How should I take Rukobia?
- Take Rukobia exactly as your healthcare provider tells you to take it.
- Take Rukobia tablets whole. Do not chew, crush, or split Rukobia tablets before swallowing.
- Take Rukobia with or without food.
- Rukobia tablets may have a slight odor (like vinegar). This is normal.
- Do not miss a dose of Rukobia. If you miss a dose of Rukobia, take it as soon as you remember. Do not take 2 doses at the same time or take more than your healthcare provider tells you to take.
- Do not run out of Rukobia. The virus in your blood may increase and the virus may become harder to treat. When your supply starts to run low, get more from your healthcare provider or pharmacy.
- If you take too much Rukobia, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Rukobia?
Rukobia can cause serious side effects including:
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having new symptoms after you start taking Rukobia.
- Heart rhythm problems (QTc prolongation). Rukobia may cause a heart rhythm problem called QTc prolongation. QTc prolongation causes an irregular heartbeat. If you are elderly, you may be at a greater risk for developing this heart problem with Rukobia.
Tell your healthcare provider right away if you feel dizzy, lightheaded, feel changes in your heartbeat, or you faint (lose consciousness). - Changes in liver function blood tests results. People with HIV-1 who take Rukobia and who also have hepatitis B or C virus infections, may be more likely to develop new or worsening changes in certain liver function blood tests during treatment with Rukobia.
- If you stop your anti-hepatitis B treatment, this could mean that your hepatitis B may become active again (reactivated). Your healthcare provider may do blood tests to check your liver during treatment with Rukobia especially if you have hepatitis B virus infection.
- Take any anti-hepatitis B or anti-hepatitis C medicines as prescribed by your healthcare provider during treatment with Rukobia.
The most common side effect of Rukobia is nausea.
These are not all the possible side effects of Rukobia.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088.
General information about the safe and effective use of Rukobia
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Rukobia for a condition for which it was not prescribed. Do not give Rukobia to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacists or healthcare provider for information about Rukobia that is written for health professionals.
How should I store Rukobia?
- Store Rukobia at room temperature between 68°F to 77°F (20°C to 25°C).
- Rukobia comes in a child-resistant package.
Keep Rukobia and all medicines out of the reach of children.
What are the ingredients in Rukobia?
Active ingredient: fostemsavir.
Inactive ingredients: colloidal silicon dioxide, hydroxypropyl cellulose, hypromellose, and magnesium stearate.
The tablet film-coating contains: iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
Label
PRINCIPAL DISPLAY PANEL
- NDC 49702-250-18
- Rukobia
- (fostemsavir)
- 600 mg
- extended-release tablets
- Rx Only
- Each film-coated tablet contains 600 mg of fostemsavir (equivalent to 725 mg fostemsavir tromethamine).
- Bottle of 60 tablets
- Note to pharmacist:
- Do not cover ALERT box with pharmacy label.
- ALERT: Find out about medicines that should NOT be taken with RUKOBIA.
- Made in UK
- Rev. 3/22
- 62000000076403


SRC: NLM .
