Ofev
Generic name: nintedanib
Drug class: Multikinase inhibitors
Medically reviewed by A Ras MD.
What is Ofev?
Ofev is a prescription medicine used to treat people with a lung disease called idiopathic pulmonary fibrosis (IPF). It is also used to treat people with a chronic (long lasting) interstitial lung disease in which lung fibrosis continues to worsen (progress). Other use of this medicine is to slow the rate of decline in lung function in people with systemic sclerosis-associated interstitial lung disease (SSc-ILD) (also known as scleroderma-associated ILD).
It is not known if Ofev is safe and effective in children.
Description
OFEV capsules contain nintedanib, a kinase inhibitor . Nintedanib is presented as the ethanesulfonate salt (esylate), with the chemical name 1H-Indole-6-carboxylic acid, 2,3-dihydro-3-[[[4-[methyl[(4-methyl-1-piperazinyl)acetyl]amino]phenyl]amino]phenylmethylene]-2-oxo-,methyl ester, (3Z)-, ethanesulfonate (1:1).
Its structural formula is:

Nintedanib esylate is a bright yellow powder with an empirical formula of C31H33N5O4∙C2H6O3S and a molecular weight of 649.76 g/mol.
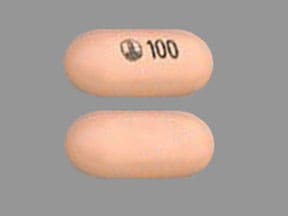
OFEV capsules for oral administration are available in 2 dose strengths containing 100 mg or 150 mg of nintedanib (equivalent to 120.40 mg or 180.60 mg nintedanib ethanesulfonate, respectively). The inactive ingredients of OFEV are the following: Fill Material: triglycerides, hard fat, lecithin. Capsule Shell: gelatin, glycerol, titanium dioxide, red ferric oxide, yellow ferric oxide, black ink.
Mechanism of Action
Nintedanib is a small molecule that inhibits multiple receptor tyrosine kinases (RTKs) and non-receptor tyrosine kinases (nRTKs). Nintedanib inhibits the following RTKs: platelet-derived growth factor receptor (PDGFR) α and β, fibroblast growth factor receptor (FGFR) 1-3, vascular endothelial growth factor receptor (VEGFR) 1-3, colony stimulating factor 1 receptor (CSF1R), and Fms-like tyrosine kinase-3 (FLT-3). These kinases except for FLT-3 have been implicated in pathogenesis of interstitial lung diseases (ILD). Nintedanib binds competitively to the adenosine triphosphate (ATP) binding pocket of these kinases and blocks the intracellular signaling cascades, which have been demonstrated to be involved in the pathogenesis of fibrotic tissue remodeling in ILD. Nintedanib also inhibits the following nRTKs: Lck, Lyn and Src kinases. The contribution of FLT-3 and nRTK inhibition to nintedanib efficacy in ILD is unknown.
What is the most important information I should know about Ofev?
- Ofev can cause birth defects or death to an unborn baby. Women should not become pregnant while taking Ofev. Women who are able to become pregnant should have a pregnancy test before starting treatment with Ofev.
- Women who are able to become pregnant should use highly effective birth control during treatment and for at least 3 months after treatment. Talk with your doctor about what birth control method is right for you during this time.
- Women using hormonal birth control should also use a barrier method of birth control (such as male condoms or spermicide).
- If you become pregnant or think you are pregnant while taking Ofev, tell your doctor right away.
What should I tell my healthcare provider before taking Ofev?
Before you take Ofev, tell your doctor about all of your medical conditions, including if you:
- have liver problems.
- have heart problems.
- have a history of blood clots.
- have a bleeding problem or a family history of a bleeding problem.
- have had recent surgery in your stomach (abdominal) area.
- are a smoker.
- are pregnant or plan to become pregnant. Ofev can harm your unborn baby. Ofev can cause birth defects or death to an unborn baby. See “What is the most important information I should know about Ofev?”
- are breastfeeding or plan to breastfeed. It is not known if Ofev passes into your breast milk. You should not breastfeed while taking Ofev.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements such as St. John’s wort. Keep a list of the medicines you take and show it to your doctor and pharmacist when you get a new medicine.
How should I take Ofev?
- Take Ofev exactly as your doctor tells you to take it.
- Your doctor will tell you how much Ofev to take and when to take it.
- Take Ofev with food. Swallow the Ofev capsules whole with a liquid.
- Do not chew or crush Ofev capsules.
- If you miss a dose of Ofev, take your next dose at your regular time. Do not take the missed dose.
- Do not take more than 300 mg of Ofev in 1 day.
- If you take too much Ofev, call your doctor or go to the nearest hospital emergency room right away.
- Your doctor should do certain blood tests before you start taking Ofev.
What are the possible side effects of Ofev?
Ofev may cause serious side effects, including:
- See “What is the most important information I should know about Ofev?”
- liver problems. Call your doctor right away if you have unexplained symptoms such as yellowing of your skin or the white part of your eyes (jaundice), dark or brown (tea colored) urine, pain on the upper right side of your stomach area (abdomen), bleeding or bruising more easily than normal, feeling tired, or loss of appetite. Your doctor will do blood tests to check how well your liver is working before starting and during your treatment with OFEV.
- diarrhea, nausea, and vomiting. While you are taking OFEV, your doctor may recommend that you drink fluids or take medicine to treat these side effects. Tell your doctor if you have diarrhea, nausea, or vomiting or if these symptoms do not go away or become worse. Tell your doctor if you are taking over-the-counter laxatives, stool softeners, and other medicines or dietary supplements that can cause diarrhea.
- heart attack. Tell your doctor right away if you have symptoms of a heart problem. These symptoms may include chest pain or pressure, pain in your arms, back, neck or jaw, or shortness of breath.
- stroke. Tell your doctor right away if you have symptoms of a stroke. These symptoms may include numbness or weakness on 1 side of your body, trouble talking, headache, or dizziness.
- bleeding problems. OFEV may increase your chances of having bleeding problems. Tell your doctor if you have unusual bleeding, bruising, or wounds that do not heal. Tell your doctor if you are taking a blood thinner, including prescription blood thinners and over-the-counter aspirin.
- tear in your stomach or intestinal wall (perforation). OFEV may increase your chances of having a tear in your stomach or intestinal wall. Tell your doctor if you have pain or swelling in your stomach area.
The most common side effects of OFEV are diarrhea, nausea, stomach pain, vomiting, liver problems, decreased appetite, headache, weight loss, and high blood pressure.
These are not all the possible side effects of OFEV. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Ofev
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Ofev for a condition for which it was not prescribed. Do not give Ofev to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about Ofev. If you would like more information, talk to your doctor. You can ask your pharmacist or doctor for information about Ofev that is written for health professionals.
For more information, go to www.ofev.com or call Boehringer Ingelheim Pharmaceuticals, Inc. at 1-800-542-6257, or (TTY) 1-800-459-9906, or scan the code below to go to www.ofev.com.
How should I store Ofev?
- Store Ofev at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep Ofev dry and protect from high heat.
Keep Ofev and all medicines out of the reach of children.
What are the ingredients in Ofev?
Active ingredient: nintedanib.
Inactive ingredients: Fill Material: triglycerides, hard fat, lecithin.
Capsule Shell: gelatin, glycerol, titanium dioxide, red ferric oxide, yellow ferric oxide, black ink.
Label
PRINCIPAL DISPLAY PANEL – 150 MG CAPSULE BOTTLE CARTON
- NDC 0597-0145-60
- Ofev®
(nintedanib
capsules) - 150 mg
- Rx only
- 60 capsules
- Boehringer
Ingelheim

PRINCIPAL DISPLAY PANEL – 100 MG CAPSULE BOTTLE CARTON
- NDC 0597-0143-60
- Ofev®
(nintedanib capsules) - 100 mg
- Rx only
- 60 capsules
- Boehringer
Ingelheim

SRC: NLM .

