Myoview
Generic name: Technetium Tc 99m Tetrofosmin
Drug class: Radiologic conjugating agents
Medically reviewed by A Ras MD.
What is Myoview used for?
Myoview is a prescription agent that is used for cardiac imaging testing.
Description
MYOVIEW 30 mL is a kit for the preparation of technetium Tc99m tetrofosmin injection for intravenous use. Technetium Tc99m tetrofosmin is a radioactive diagnostic agent. Each multiple-dose 30 mL glass vial contains a sterile, non-pyrogenic, lyophilized powder of 1.38 mg tetrofosmin [6,9-bis(2-ethoxyethyl)-3,12-dioxa-6,9-diphosphatetradecane], 0.09 mg stannous chloride dihydrate, (minimum stannous tin 0.015 mg; total stannous and stannic tin 0.0522 mg) 1.92 mg disodium sulphosalicylate, 3 mg sodium D-gluconate, and 11 mg sodium hydrogen carbonate, and 3 mg ascorbic acid. The lyophilized powder is sealed under a nitrogen atmosphere with a rubber closure. The product contains no antimicrobial preservative. The chemical formula of tetrofosmin is C18H40O4P2with the following structural formula:
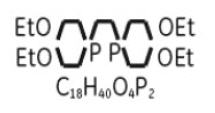
When sterile, pyrogen-free sodium pertechnetate Tc99m in isotonic saline is added to the vial, a Tc99m complex of tetrofosmin is formed. The reconstituted product is a clear solution and the pH is in the range of 7.5-9.0.
Physical Characteristics
Technetium Tc99m decays by isomeric transition with a physical half-life of 6 hours. Photons that are useful for imaging studies are listed in Table 2.
| Radiation | Mean % disintegration | Mean energy (keV) |
|---|---|---|
| Gamma 2 | 88.5 | 140.5 |
11.3 External Radiation
The air-kerma-rate (exposure-rate) constant for technetium Tc99m is 5.23 m2∙pGy∙(MBq)–1∙s–1 [0.795 cm2∙R∙(mCi)–1∙h–1].
A range of values for the relative radiation attenuation by various thicknesses of Pb shielding is shown in Table 3. For example, the use of 3mm thick Pb will decrease the external radiation exposure by a factor of approximately 1000.
| Shield thickness (Pb) mm | Factor of attenuation |
|---|---|
| 0.25 | 0.5 |
| 1 | 10-1 |
| 2 | 10-2 |
| 3 | 10-3 |
| 4 | 10-4 |
| 5 | 10-5 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals relative to the time of calibration are shown in Table 4.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
|
|||
| 0* | 1.000 | 7 | 0.446 |
| 1 | 0.891 | 8 | 0.397 |
| 2 | 0.794 | 9 | 0.354 |
| 3 | 0.707 | 10 | 0.315 |
| 4 | 0.630 | 11 | 0.281 |
| 5 | 0.562 | 12 | 0.250 |
| 6 | 0.500 | 24 | 0.063 |
Mechanism of Action
Technetium (99mTc) tetrofosmin is a lipophilic, cationic complex which diffuses passively through the cell membrane and is locally retained actively due to the presence of intact mitochondria reflecting the presence of viable cells. After intravenous injection, it is distributed within the myocardium according to myocardial perfusion and viability.
Before taking Myoview, tell your doctor:
- If you are allergic to Myoview; any part of this medicine; or any other drugs, foods, or substances. Tell your doctor about the allergy and what signs you had.
- If you are breast-feeding. Do not breast-feed for 60 hours after getting Myoview.
This medicine may interact with other drugs or health problems.
Tell your doctor and pharmacist about all of your drugs (prescription or OTC, natural products, vitamins) and health problems. You must check to make sure that it is safe for you to take Myoview with all of your drugs and health problems. Do not start, stop, or change the dose of any drug without checking with your doctor.
What are some things I need to know or do while I take Myoview?
- Tell all of your health care providers that you take Myoview. This includes your doctors, nurses, pharmacists, and dentists.
- This medicine is radioactive. You will need to follow what the doctor has told you to lessen being exposed to Myoview.
- Drink lots of noncaffeine liquids after using Myoview unless told to drink less liquid by your doctor.
- You will need to empty your bladder often after the test is over as your doctor has told you.
- Tell your doctor if you are pregnant or plan on getting pregnant. You will need to talk about the benefits and risks of using Myoview while you are pregnant.
How is Myoview best taken?
Use Myoview as ordered by your doctor. Read all information given to you. Follow all instructions closely.
- It is given as a shot into a vein.
What do I do if I miss a dose?
- Call your doctor to find out what to do.
What are the side effects of Myoview that I need to call my doctor about immediately?
WARNING/CAUTION: Even though it may be rare, some people may have very bad and sometimes deadly side effects when taking a drug. Tell your doctor or get medical help right away if you have any of the following signs or symptoms that may be related to a very bad side effect:
- Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing, swallowing, or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat.
- Chest pain or pressure or a fast heartbeat.
- A heartbeat that does not feel normal.
- Very bad dizziness or passing out.
- Shortness of breath.
- A burning, numbness, or tingling feeling that is not normal.
- Seizures.
- Feeling sleepy.
- Cough.
- Very bad belly pain.
- Weakness on 1 side of the body, trouble speaking or thinking, change in balance, drooping on one side of the face, or blurred eyesight.
What are some other side effects of Myoview?
All drugs may cause side effects. However, many people have no side effects or only have minor side effects. Call your doctor or get medical help if any of these side effects or any other side effects bother you or do not go away:
- Flushing.
- Headache.
- Belly pain.
- Dizziness.
These are not all of the side effects that may occur. If you have questions about side effects, call your doctor. Call your doctor for medical advice about side effects.
You may report side effects to the FDA at 1-800-332-1088. You may also report side effects at https://www.fda.gov/medwatch.
If overdose is suspected:
If you think there has been an overdose, call your poison control center or get medical care right away. Be ready to tell or show what was taken, how much, and when it happened.
How do I store and/or throw out Myoview?
- If you need to store Myoview at home, talk with your doctor, nurse, or pharmacist about how to store it.
Label
PRINCIPAL DISPLAY PANEL – KIT LABEL
- GE Healthcare
- Rx ONLY
- Sterile
Pyrogen-free
Diagnostic - For Intravenous
administration only. - MYOVIEW™
30mL - Kit for the Preparation
of Technetium Tc99m
Tetrofosmin for
Injection - 5 multi-dose
vial pack - NDC 17156-026-30
- Contents of Kit
5 STERILE REACTION VIALS
Each vial contains lyophilized form of:
1.38 mg Tetrofosmin
0.09 mg Stannous chloride dihydrate
1.92 mg Disodium sulphosalicylate
3.0 mg Sodium D-gluconate
11.0 mg Sodium hydrogen carbonate
3.0 mg Ascorbic Acid
5 PRESSURE SENSITIVE LABELS
1 PACKAGE INSERT - Myoview is a trademark of GE Healthcare.
- GE and the GE Monogram are trademarks
of General Electric Company. - Manufactured for:
GE Healthcare
Medi-Physics Inc.
Arlington Heights, IL 60004 - by: GE Healthcare AS, Oslo, Norway
- EXP.:
- LOT:

SRC: NLM .
