Lysteda
Generic name: tranexamic acid (Lysteda)
Drug class: Miscellaneous coagulation modifiers
Medically reviewed by A Ras MD.
What is Lysteda?
Lysteda is a prescription medicine used to treat your heavy monthly period (menstruation) when your bleeding gets in the way of social, leisure and physical activities. Lysteda does not contain any hormones. On average, Lysteda has been shown to lower the amount of blood lost during your monthly period by about one-third, but it is not meant to stop your period.
Lysteda is taken only during your period and is not meant to treat pre-menstrual symptoms (symptoms that occur before your bleeding starts). Lysteda does not affect your fertility and cannot be used as birth control. Lysteda does not protect you against diseases that you may get if you have unprotected sex.
Lysteda has not been studied in adolescents younger than 18 years of age.
Lysteda is not for women who have already gone through menopause (post-menopausal).
Description
LYSTEDA is an antifibrinolytic drug administered orally. The chemical name is trans-4-aminomethyl-cyclohexanecarboxylic acid. The structural formula is:
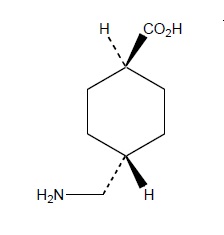
Tranexamic acid is a white crystalline powder. It is freely soluble in water and in glacial acetic acid and is very slightly soluble in ethanol and practically insoluble in ether. The molecular formula is C8H15N02 and the molecular weight is 157.2.
LYSTEDA is provided as white oval-shaped tablets and are not scored. Each tablet is debossed with the marking “FP650.” The active ingredient in each tablet is 650 mg tranexamic acid. The inactive ingredients contained in each tablet are: microcrystalline cellulose, colloidal silicon dioxide, pregelatinized corn starch, povidone, hypromellose, stearic acid, and magnesium stearate.
Mechanism of Action
Tranexamic acid is a synthetic lysine amino acid derivative, which diminishes the dissolution of hemostatic fibrin by plasmin. In the presence of tranexamic acid, the lysine receptor binding sites of plasmin for fibrin are occupied, preventing binding to fibrin monomers, thus preserving and stabilizing fibrin’s matrix structure.
The antifibrinolytic effects of tranexamic acid are mediated by reversible interactions at multiple binding sites within plasminogen. Native human plasminogen contains 4 to 5 lysine binding sites with low affinity for tranexamic acid (Kd = 750 μmol/L) and 1 with high affinity (Kd = 1.1 μmol/L). The high affinity lysine site of plasminogen is involved in its binding to fibrin. Saturation of the high affinity binding site with tranexamic acid displaces plasminogen from the surface of fibrin. Although plasmin may be formed by conformational changes in plasminogen, binding to and dissolution of the fibrin matrix is inhibited.
Who should not take Lysteda?
Do not take Lysteda if you:
- Are using a form of birth control that contains estrogen and a progestin (like a birth control pill, patch, or vaginal ring). Ask your healthcare provider before taking Lysteda if you are not sure if your birth control method contains estrogen and a progestin.
- Currently have a blood clot
- Have ever had a blood clot
- Have been told that you are at risk of having a blood clot
- Are allergic to Lysteda or tranexamic acid
What should I tell my healthcare provider before taking Lysteda?
Before taking Lysteda, tell your healthcare provider about all of your medical conditions, including whether:
- You have ever had a blood clot or been told that you are at risk of having a blood clot
- You are using a form of birth control that contains estrogen and a progestin (like a birth control pill, patch, or vaginal ring). Using hormonal birth control along with Lysteda may increase your chance of having a serious blood clot, stroke, or heart attack. For this reason, do not use Lysteda if you use a form of birth control that contains estrogen and a progestin.
- You are pregnant or think you may be pregnant
- You are breastfeeding or plan to breast-feed. Lysteda can pass into your milk. Talk to your healthcare provider about the best way to feed your baby if you take Lysteda.
- The time between the start of your periods is less than 21 days or more than 35 days
- You have any other medical conditions
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Lysteda and other medicines can affect each other, causing side effects. Lysteda can affect the way other medicines work and other medicines can affect how Lysteda works.
Especially tell your healthcare provider if you take:
- Birth control pills or other hormonal birth control
- Medicines used to help your blood form clots
- Medicines used to break up blood clots
- Any medicines to treat leukemia
Ask your healthcare provider if you are not sure if your medicine is one that is described above.
How should I take Lysteda?
- Take Lysteda exactly as your healthcare provider tells you.
- Do not take Lysteda until your period has started.
- Do not take Lysteda for more than 5 days in a row.
- Do not take Lysteda when you do not have your period.
- Once your period has started, take 2 tablets of Lysteda three times per day (e.g., in the morning, afternoon, and evening).
- Lysteda tablets should be swallowed whole and not chewed or broken apart.
- Lysteda may be taken with or without food.
- Do not take more than 6 tablets of Lysteda in a day. If you take more than 6 tablets, call your healthcare provider.
- If you miss a dose, take it when you remember, and then take your next dose at least six hours later. Do not take more than two tablets at a time to make up for missed doses.
- If Lysteda does not help to lessen bleeding with your periods after 2 cycles or seems to stop working, talk to your healthcare provider.
What are the possible side effects of Lysteda?
Lysteda can cause serious side effects, including:
- Blood clots. You may have a higher risk of having serious blood clots if you take Lysteda with:
- medicines used to help your blood form clots
- some medicines used to treat leukemia
- Eye changes. Stop taking Lysteda and promptly report any eye problems you have while taking Lysteda. Your doctor will refer you to an eye doctor who will examine your eyes.
- Allergic reaction. If you have severe shortness of breath and your throat feels tight, stop taking Lysteda and get medical care right away.
The most common side effects of Lysteda include:
- Headaches
- Sinus and nasal problems
- Back pain
- nausea
- Pain in your abdomen
- Pain in your muscles or joints
- Anemia
- Fatigue
Tell your healthcare provider if you have any side effect that bothers you or does not go away.
These are not all of the possible side effects of Lysteda. For more information, ask your healthcare provider or pharmacist.
If you notice a change in your usual bleeding pattern that worries you, or your heavy bleeding continues, contact your healthcare provider right away. This may be a sign of a more serious condition.
Call your healthcare provider for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088. You may also report side effects to Ferring Pharmaceuticals Inc. at 1-888-FERRING (1-888-337-7464).
General information about the safe and effective use of Lysteda
Medicines are sometimes prescribed for conditions that are not mentioned in Patient Information Leaflets. Do not use Lysteda for a condition for which it was not prescribed. Do not give Lysteda to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about Lysteda. If you would like more information about Lysteda, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Lysteda that is written for healthcare professionals. For more information, go to www.lysteda.com or call 1-888-FERRING (1-888-337-7464).
How should I store Lysteda?
Store Lysteda at room temperature between 59°F to 86°F (15°C to 30°C).
Keep Lysteda and all medicines out of the reach of children.
What are the ingredients in Lysteda?
Active ingredient: tranexamic acid
Inactive ingredients: microcrystalline cellulose, colloidal silicon dioxide, pregelatinized corn starch, povidone, hypromellose, stearic acid, and magnesium stearate.
Label
PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL – 650 mg Tablet Bottle Label
- Rx only
NDC 69918-300-30 - Lysteda®
(tranexamic acid, USP) tablets - 650 mg
30 tablets


SRC: NLM .
