Cortifoam
Generic name: hydrocortisone rectal (foam, enema)
Brand names: Cortenema, Cortifoam
Drug class: Glucocorticoids
Medically reviewed by A Ras MD.
What is Cortifoam?
Cortifoam is a prescription medicine that is used to treat ulcerative colitis.
Description
Cortifoam® (hydrocortisone acetate rectal aerosol) 10% Rectal Foam contains hydrocortisone acetate 10% in a base containing propylene glycol, emulsifying wax, polyoxyethylene-10-stearyl ether, cetyl alcohol, methylparaben, propylparaben, trolamine, purified water and inert propellants: isobutane and propane.
Each application delivers approximately 900 mg of foam containing 80 mg of hydrocortisone (90 mg of hydrocortisone acetate).
The molecular weight of hydrocortisone acetate is 404.50. It is designated chemically as pregn-4-ene-3,20-dione,21-(acetyloxy)-11,17-dihydroxy-,(11β)-. The empirical formula is C23H32O6 and the structural formula is:

Hydrocortisone acetate, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is insoluble in water (1 mg/100 mL) and slightly soluble in alcohol and chloroform.
Before taking Cortifoam, tell your doctor:
- If you are allergic to Cortifoam; any part of this medicine; or any other drugs, foods, or substances. Tell your doctor about the allergy and what signs you had.
- If you have any of these health problems: Bowel block; fistula; a fungal infection; holes, sores, or wounds on the rectal area; peritonitis; or recent bowel surgery.
This is not a list of all drugs or health problems that interact with this medicine.
Tell your doctor and pharmacist about all of your drugs (prescription or OTC, natural products, vitamins) and health problems. You must check to make sure that it is safe for you to take Cortifoam with all of your drugs and health problems. Do not start, stop, or change the dose of any drug without checking with your doctor.
What are some things I need to know or do while I take Cortifoam?
- Tell all of your health care providers that you take Cortifoam. This includes your doctors, nurses, pharmacists, and dentists.
- Talk with your doctor before you use other drugs or products on your skin.
- Do not put on cuts, scrapes, or damaged skin.
- Use care when putting on a large part of the skin or where there are open wounds. Talk with the doctor.
- This medicine may cause harm if swallowed. If Cortifoam is swallowed, call a doctor or poison control center right away.
- Do not use Cortifoam for longer than you were told by your doctor.
- Use with care in children. Talk with the doctor.
- This medicine may affect growth in children and teens in some cases. They may need regular growth checks. Talk with the doctor.
- Tell your doctor if you are pregnant, plan on getting pregnant, or are breast-feeding. You will need to talk about the benefits and risks to you and the baby.
How is Cortifoam best taken?
Use Cortifoam as ordered by your doctor. Read all information given to you. Follow all instructions closely.
- Use as you have been told, even if your signs get better.
- This medicine is for rectal use only. Do not take Cortifoam by mouth.
- Wash your hands before and after use.
- Shake well before use.
- Use an applicator to put in foam. Do not put the foam container right into the rectum.
- This medicine may catch on fire. Do not use near an open flame or while smoking.
What do I do if I miss a dose?
- Use a missed dose as soon as you think about it.
- If it is close to the time for your next dose, skip the missed dose and go back to your normal time.
- Do not use 2 doses at the same time or extra doses.
What are the side effects of Cortifoam that I need to call my doctor about immediately?
WARNING/CAUTION: Even though it may be rare, some people may have very bad and sometimes deadly side effects when taking a drug. Tell your doctor or get medical help right away if you have any of the following signs or symptoms that may be related to a very bad side effect:
- Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing, swallowing, or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat.
- Signs of high blood sugar like confusion, feeling sleepy, more thirst, more hungry, passing urine more often, flushing, fast breathing, or breath that smells like fruit.
- Signs of a weak adrenal gland like a very bad upset stomach or throwing up, very bad dizziness or passing out, muscle weakness, feeling very tired, mood changes, not hungry, or weight loss.
- Signs of Cushing’s disease like weight gain in the upper back or belly, moon face, very bad headache, or slow healing.
- Change in eyesight.
- Very bad headache.
- Irritation where Cortifoam is used.
- Very bad bleeding from rectum or rectal pain.
What are some other side effects of Cortifoam?
All drugs may cause side effects. However, many people have no side effects or only have minor side effects. Call your doctor or get medical help if any of these side effects or any other side effects bother you or do not go away:
- Stinging.
- Dryness.
- Burning.
- Tingling.
These are not all of the side effects that may occur. If you have questions about side effects, call your doctor. Call your doctor for medical advice about side effects.
You may report side effects to the FDA at 1-800-332-1088. You may also report side effects at https://www.fda.gov/medwatch.
If overdose is suspected:
If you think there has been an overdose, call your poison control center or get medical care right away. Be ready to tell or show what was taken, how much, and when it happened.
How do I store and/or throw out Cortifoam?
- Store at room temperature. Do not freeze.
- Protect from heat or open flame. Do not puncture or burn even if it seems empty.
- Keep all drugs in a safe place. Keep all drugs out of the reach of children and pets.
- Throw away unused or expired drugs. Do not flush down a toilet or pour down a drain unless you are told to do so. Check with your pharmacist if you have questions about the best way to throw out drugs. There may be drug take-back programs in your area.
Label
PRINCIPAL DISPLAY PANEL – 15 G LABEL
- NDC 0037-6830-15
- cortifoam®
(hydrocortisone acetate 10%) - rectal foam
- Rx Only
- 15 g net wt
DIRECTIONS FOR USE
|
APPLICATOR |
FOAM CONTAINER |
||
 |
 |
||
|
1 Shake foam container vigorously for 5 – 10 seconds before each use. Do not remove container cap during use of the product. |
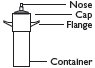
2 Hold container upright on a level surface and gently place the tip of the applicator onto the nose of the container cap. |
||
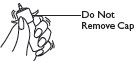 |
CONTAINER MUST BE HELD UPRIGHT TO OBTAIN PROPER FLOW OF MEDICATION. |
||
 |
|||
|
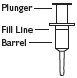
3 Pull plunger past the fill line on the applicator barrel. |
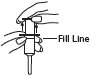 |
||
|
4 To fill applicator barrel, press down firmly on cap flanges, hold for 1 – 2 seconds and release. Pause 5 – 10 seconds to allow foam to expand in applicator barrel. Repeat until foam reaches fill line. Remove applicator from container cap. Allow some foam to remain on the applicator tip. |
|||
|
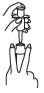
5 Hold applicator firmly by barrel, making sure thumb and middle finger are positioned securely underneath and resting against barrel wings. place index finger over the plunger. Gently insert tip into anus. Once in place, push plunger to expel foam, then withdraw applicator. |
|||
|
6 After each use, applicator parts should be pulled apart for thorough cleaning with warm water. The container cap and underlying tip should also be pulled apart and rinsed to help prevent buildup of foam and possible blockage. |
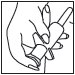 |
||
Description: cortifoam® (hydrocortisone acetate) 10% rectal foam contains hydrocortisone acetate 10% in a base containing propylene glycol, emulsifying wax, polyoxyethylene-10-stearyl ether, cetyl alcohol, methylparaben, propyl-paraben, trolamine, purified water and inert propellants: isobutane and propane.
Dosage: One applicatorful once or twice daily for two or three weeks, and every second day thereafter, administered rectally. See back of carton for full directions for use.
Please see package insert for further information. For rectal use only.
117610-0820 UC-683015-04
Caution: Do not insert any part of the aerosol container directly into the anus. Apply to anus only with the enclosed applicator.
WARNINGS: Contents of the container are under pressure. Do not burn or puncture the aerosol container. Do not store at temperatures above 120°F (49°C).
KEEP OUT OF REACH OF CHILDREN.
Store upright at controlled room temperature 20° – 25°C (68° – 77°F).
DO NOT REFRIGERATE
Distributed by: Meda Pharmaceuticals Inc., Somerset, New Jersey 08873-4120
©2020 Mylan Specialty L.P.
Cortifoam is a registered trademark of Alaven Pharmaceutical LLC, a Mylan company.

SRC: NLM .
