Eucrisa
Generic name: crisaborole topical
Drug class: Miscellaneous topical agents
Medically reviewed by A Ras MD.
What is Eucrisa?
Eucrisa is a prescription medicine used on the skin (topical) to treat mild to moderate eczema (atopic dermatitis) in adults and children 3 months of age and older.
It is not known if Eucrisa is safe and effective in children under 3 months of age.
Description
EUCRISA contains 2% crisaborole (w/w) in a petrolatum-based, white to off-white ointment and is for topical use. The active ingredient, crisaborole, is a phosphodiesterase-4 (PDE-4) inhibitor.
Crisaborole is described chemically as 5-(4-cyanophenoxy)-1,3-dihydro-1-hydroxy-[2,1]-benzoxaborole. The empirical formula is C14H10BNO3 and the molecular weight is 251.1 g/mol.
The structural formula is represented below:
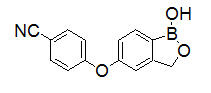
Crisaborole drug substance is freely soluble in common organic solvents such as isopropyl alcohol and propylene glycol, and insoluble in water.
Each gram of EUCRISA contains 20 mg of crisaborole in an ointment containing white petrolatum, propylene glycol, mono- and di-glycerides, paraffin, butylated hydroxytoluene, and edetate calcium disodium.
Mechanism of Action
Crisaborole is a phosphodiesterase 4 (PDE-4) inhibitor. PDE-4 inhibition results in increased intracellular cyclic adenosine monophosphate (cAMP) levels. The specific mechanism(s) by which crisaborole exerts its therapeutic action for the treatment of atopic dermatitis is not well defined.
What is the most important information I should know about Eucrisa?
Eucrisa is for use on skin (topical use) only. Do not use Eucrisa in your eyes, mouth, or vagina.
Who should not use Eucrisa?
Do not use Eucrisa if you are allergic to crisaborole or any of the ingredients in Eucrisa. See the end of this guide for a complete list of ingredients in Eucrisa.
What should I tell my healthcare provider before taking Eucrisa?
Before using Eucrisa, tell your healthcare provider about all of your medical conditions, including if you:
- are pregnant or plan to become pregnant. It is not known if Eucrisa will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Eucrisa passes into your breast milk.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I use Eucrisa?
- Use Eucrisa exactly as your healthcare provider tells you to use it.
- Apply a thin layer of Eucrisa to the affected areas 2 times each day.
- Wash your hands after applying Eucrisa, unless hands are being treated. If someone else applies Eucrisa for you, they should wash their hands after applying Eucrisa.
What are the possible side effects of Eucrisa?
Eucrisa may cause side effects.
- Allergic reactions. Eucrisa may cause allergic reactions at or near the application site. These can be seriousand may include hives, itching, swelling, redness. If you have any of these symptoms, stop using Eucrisa and get medical help right away.
The most common side effect of Eucrisa is application site pain, such as burning or stinging.
These are not the only possible side effect of Eucrisa.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Eucrisa
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information guide. Do not use Eucrisa for a condition for which it was not prescribed. Do not give Eucrisa to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Eucrisa that is written for healthcare professionals.
How should I store Eucrisa?
- Store Eucrisa at room temperature, between 68°F and 77°F (20°C and 25°C).
- Keep the tube tightly closed.
What are the ingredients in Eucrisa?
Active ingredient: crisaborole
Inactive ingredients: white petrolatum, propylene glycol, mono- and di-glycerides, paraffin, butylated hydroxytoluene, and edetate calcium disodium.
Label
PRINCIPAL DISPLAY PANEL – 60 G TUBE LABEL
- eucrisa®
(crisaborole) ointment 2% - NDC 55724-211-21
60 g
Rx only - For Topical Use Only.
- Not for ophthalmic, oral, or intravaginal use.

SRC: NLM .
